Elements
Elements are pure substances that consist of only one type of atom, distinguished by the number of protons in their nuclei.
Physical Properties
- Atomic Structure: Each element is defined by its atomic number, the number of protons in its nucleus.
- States of Matter: Elements can exist in solid, liquid, or gaseous states under standard conditions. For instance, carbon is solid, while helium is a gas.
- Melting and Boiling Points: Elements have specific melting and boiling points. For example, iron melts at 1538°C.
- Density: The density of each element is unique, like the high density of lead compared to lithium.
Chemical Properties
- Reactivity: The reactivity of elements varies greatly. For example, fluorine is highly reactive, while neon is almost completely inert.
- Valency: The valency of an element determines how it bonds with other atoms. Oxygen, with a valency of 2, forms two bonds.
Composition and Examples
- Pure Substance: An element is a pure chemical substance composed of atoms with the same number of protons.
- Examples: Gold (Au), known for its lustrous yellow appearance and inert nature, and Oxygen (O), essential for respiration.
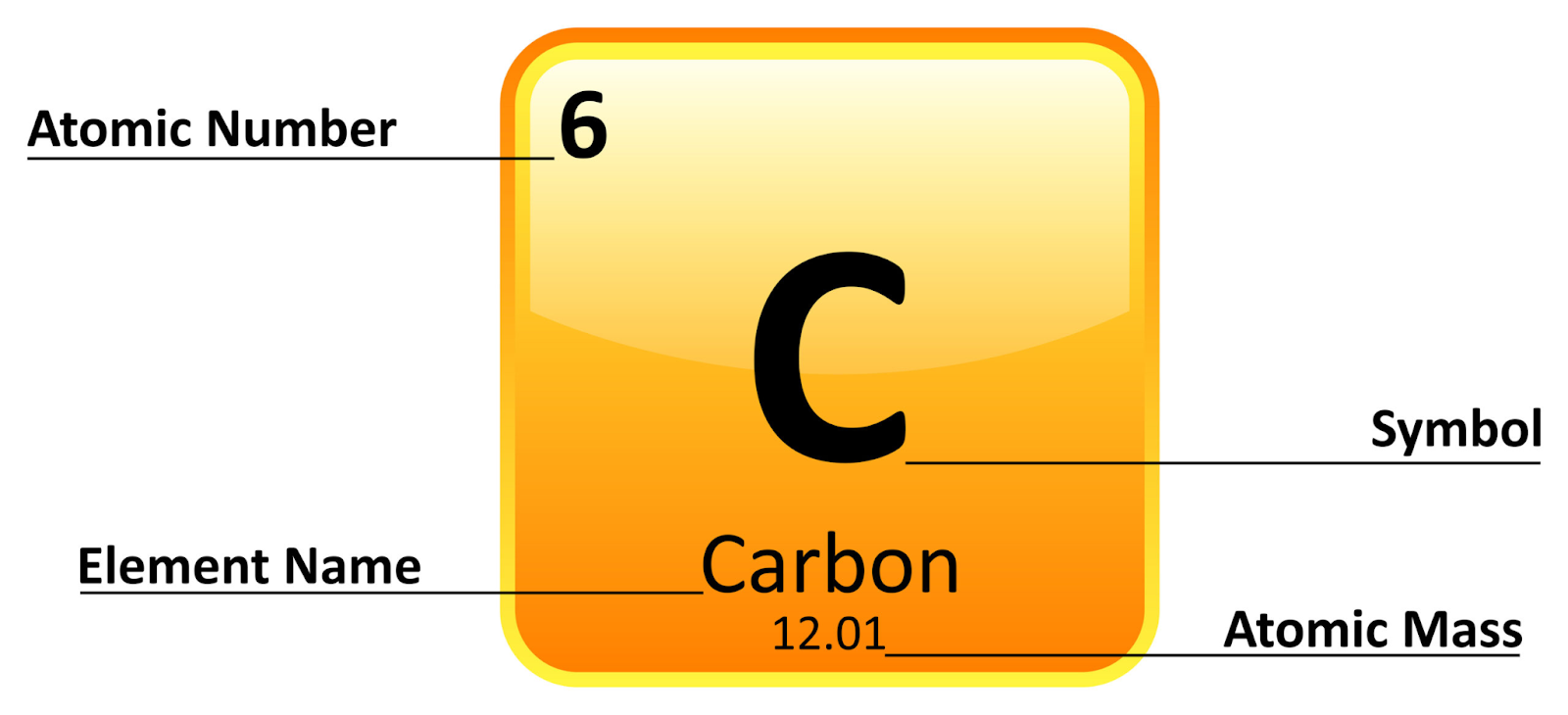
Image courtesy of Science Sparks
Compounds
Compounds are substances formed by the chemical combination of two or more elements in fixed proportions, held together by chemical bonds.
Physical Properties
- Fixed Composition: The atoms in a compound are combined in a fixed ratio. Water, for example, always has two hydrogen atoms for every oxygen atom.
- Distinctive Properties: The properties of a compound differ from those of its constituent elements. Sodium chloride (table salt) is edible, while neither sodium nor chlorine is safe to consume in their elemental forms.
- Solubility: Compounds differ in solubility. Sodium chloride is soluble in water, while calcium carbonate is not.
Chemical Properties
- Chemical Bonds: The formation of compounds involves the creation of ionic or covalent bonds between atoms. Sodium chloride forms through ionic bonding, while water forms through covalent bonding.
- Chemical Reactions: Compounds can undergo chemical reactions, breaking bonds and forming new substances.
Composition and Examples
- Molecular Structure: A compound consists of molecules formed from atoms of different elements.
- Examples: Carbon dioxide (CO₂) is a compound consisting of one carbon atom double-bonded to two oxygen atoms. It is a key component in photosynthesis and respiration.

Image courtesy of YourDictionary
Mixtures
Mixtures are physical combinations of two or more substances (elements or compounds) where each substance retains its chemical identity and properties.
Physical Properties
- Variable Composition: Unlike compounds, mixtures do not have a fixed ratio of components. Air, for example, consists of nitrogen, oxygen, and other gases in varying proportions.
- Separation Techniques: Mixtures can be separated into their components by physical methods. Filtration can separate a mixture of sand and water, while distillation can separate alcohol from water.
- No Definite Melting/Boiling Points: Mixtures do not have consistent melting or boiling points. The boiling point of a mixture of ethanol and water varies depending on their ratio.
Chemical Properties
- Retention of Properties: In a mixture, each component retains its original physical and chemical properties. In a saltwater solution, both salt and water retain their individual properties.
- No Chemical Formula: Unlike compounds, mixtures do not have a chemical formula.
Composition and Examples
- Types of Mixtures: Mixtures can be either homogeneous (uniform composition throughout) like saltwater, or heterogeneous (non-uniform composition) like a mixture of oil and water.
- Examples: Granite is a heterogeneous mixture of minerals like quartz, feldspar, and mica.
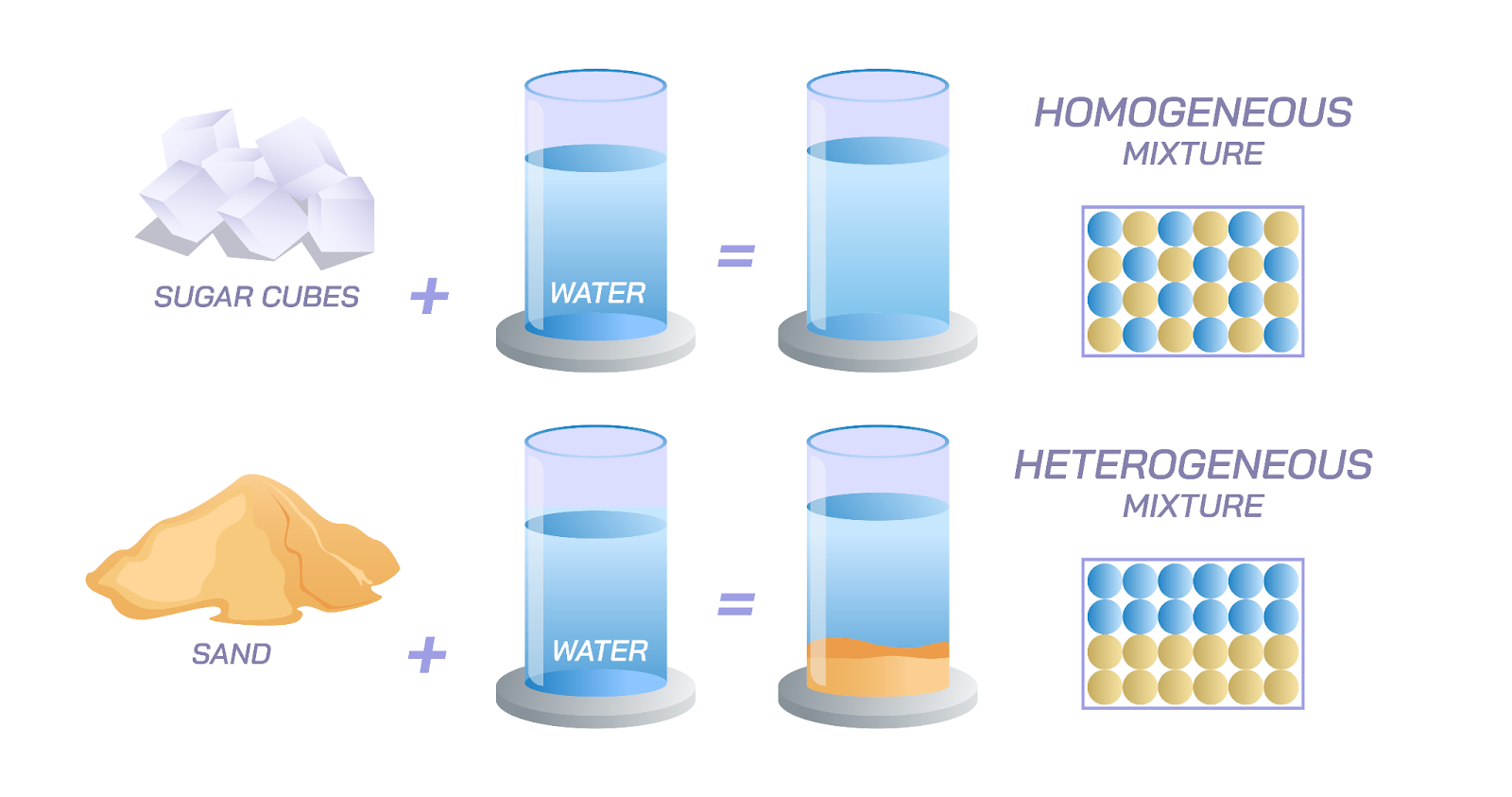
Image courtesy of Anshuman
Comparison and Summary
Elements
- Simplest Form: Elements are the simplest form of matter and cannot be broken down further.
- Unique Identity: Each element has a unique set of properties and a symbol on the Periodic Table.
Compounds
- Chemical Combination: Compounds result from the chemical bonding of elements in fixed ratios.
- New Properties: The properties of a compound are different from those of its constituent elements.
Mixtures
- Physical Blends: Mixtures are formed by physically blending two or more substances.
- Retained Properties: Components in a mixture retain their original properties.
Key Differences
- Elements are pure substances with unique properties.
- Compounds are chemical combinations of elements with new properties.
- Mixtures are physical blends without chemical bonding and retain the properties of their constituents.
In-depth knowledge of these differences is vital for understanding the nature of substances encountered in chemistry. This foundation enables students to progress to more advanced topics, such as chemical reactions and material science, which are integral parts of the IGCSE Chemistry syllabus.
FAQ
The melting and boiling points of mixtures generally vary and are not as defined as those of pure substances. In a pure substance, whether an element or a compound, the melting and boiling points are specific and characteristic of that substance. For example, pure water boils at 100°C under standard atmospheric pressure. However, mixtures have varying melting and boiling points because they contain more than one component, and each component may have different melting and boiling points. In mixtures like alloys or solutions, the presence of different substances alters the boiling and melting points from those of the pure components. For instance, seawater (a mixture of salt and water) has a lower freezing point than pure water. This variability is due to the interactions between the different components in the mixture.
Mixtures cannot be represented by a chemical formula because they do not have a fixed composition or defined molecular structure, unlike compounds. A chemical formula represents the exact number and type of atoms in a molecule of a compound, signifying a fixed ratio and a consistent chemical structure. For instance, water is always H₂O, indicating two hydrogen atoms bonded to one oxygen atom. Mixtures, however, are physical combinations of different substances where each retains its individual chemical properties and identity. For example, air is a mixture of various gases like nitrogen, oxygen, carbon dioxide, and others in varying proportions. Since the ratio of these gases is not fixed and they are not chemically bonded, air cannot be represented by a single chemical formula.
Separating the components of a mixture is generally easier than decomposing a compound due to the nature of the bonds involved. In mixtures, the components are physically combined but not chemically bonded. This means that each substance in a mixture retains its chemical identity and can be separated by physical methods like filtration, distillation, or evaporation, which rely on physical properties like boiling points or solubility. For instance, a mixture of sand and water can be easily separated by filtration. However, decomposing a compound requires breaking the chemical bonds that hold the atoms together, which typically involves a chemical reaction. This process is usually more complex and requires specific conditions like heat, light, or a catalyst. For example, breaking down water into hydrogen and oxygen requires an electric current in a process known as electrolysis, which is more complex than the physical separation of a mixture.
Chemically changing an element into a compound involves a chemical reaction where the element reacts with another element or compound, forming a new substance with different properties. For instance, hydrogen gas (an element) can react with oxygen gas (another element) to form water (a compound). This reaction involves the breaking and forming of chemical bonds and results in a substance with entirely different properties from the original elements. Turning an element into a mixture, however, is more about physical blending than chemical change. For example, mixing powdered iron (an element) with sulfur powder (another element) results in a mixture where both elements retain their individual properties. The key difference is that in forming compounds, chemical bonds are made or broken, while in mixtures, the substances are just physically combined.
Physical changes in elements, compounds, and mixtures primarily involve changes in state or appearance without altering the chemical composition. For elements, a physical change might be the transition from solid to liquid (melting) or liquid to gas (vaporisation), as seen in pure substances like iron or nitrogen. These changes are reversible and depend on temperature and pressure. In compounds, physical changes also include phase changes. For example, water (a compound) can evaporate into steam or freeze into ice, but its molecular structure (H₂O) remains unchanged. Mixtures, however, exhibit physical changes unique to their heterogeneous composition. For example, a mixture of ice and salt demonstrates a depression in the freezing point of water. In mixtures, the individual components each respond according to their properties, and separation methods like filtration or distillation exploit these differences.
Practice Questions
Compounds and mixtures can be distinguished by their composition, properties, and the methods used to separate them. A compound is a substance formed when two or more elements are chemically bonded together in fixed proportions, displaying properties different from those of its constituent elements. For example, water (H₂O) is a compound where hydrogen and oxygen are chemically bonded, and it has unique properties like a specific boiling point. On the other hand, a mixture is a blend of two or more substances where each retains its original properties, and the components can be physically separated. For instance, air is a mixture of gases like nitrogen, oxygen, and others in variable proportions and can be separated by methods like fractional distillation.
In the given list, gold (Au) is an element. It is a pure substance consisting of only one type of atom, and it cannot be broken down into simpler substances. Carbon dioxide (CO₂) is a compound, formed from chemically bonding carbon and oxygen in a fixed ratio, resulting in properties different from those of the individual elements. Saltwater, a blend of salt (sodium chloride) and water, is a mixture. In this mixture, the components (salt and water) retain their individual properties and can be separated through physical processes such as evaporation or distillation.

