In A-Level Chemistry, a profound understanding of the gaseous state is essential. This section delves into the dynamics of gas pressure and its intrinsic link with particle collisions, grounded in the principles of the kinetic theory of gases.
Introduction to Gas Pressure
Gas pressure is an intrinsic property of gases, originating from the force exerted by gas molecules upon the walls of their container. This concept is pivotal in understanding the behaviour of gases in various conditions.
Fundamentals of Gas Pressure:
- Origin of Pressure: The pressure exerted by a gas is a direct result of the momentum transfer during the collision of gas particles with the container walls.
- Collision Frequency: The number of these collisions per unit time is proportional to the pressure exerted by the gas.
- Temperature Dependence: An increase in temperature leads to an increase in the kinetic energy of gas particles, resulting in more forceful collisions and, consequently, higher gas pressure.
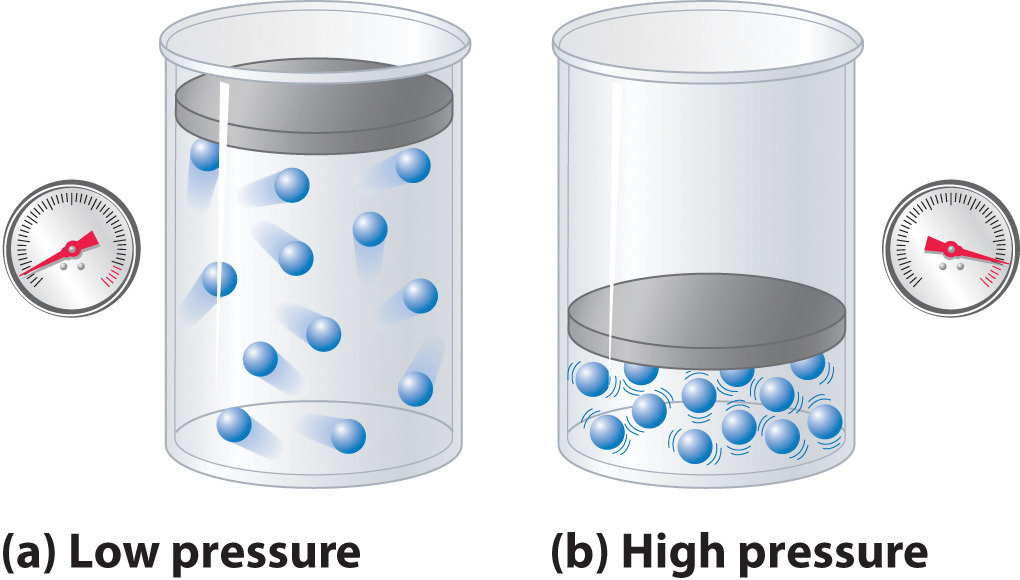
Image courtesy of Save My Exams
Kinetic Theory of Gases
The kinetic theory offers a detailed molecular-level explanation for the properties and behaviours of gases, including pressure, volume, and temperature relationships.
Principles of the Kinetic Theory:
- Random and Constant Motion: Gas particles are in a state of continuous, random motion. This motion leads to collisions among particles and with the container walls.
- Nature of Collisions: These collisions are considered perfectly elastic, meaning they do not result in a loss of kinetic energy.
- Macroscopic and Microscopic Views: The theory provides a bridge between the observable, macroscopic behaviours of gases and their microscopic, molecular-level activities.
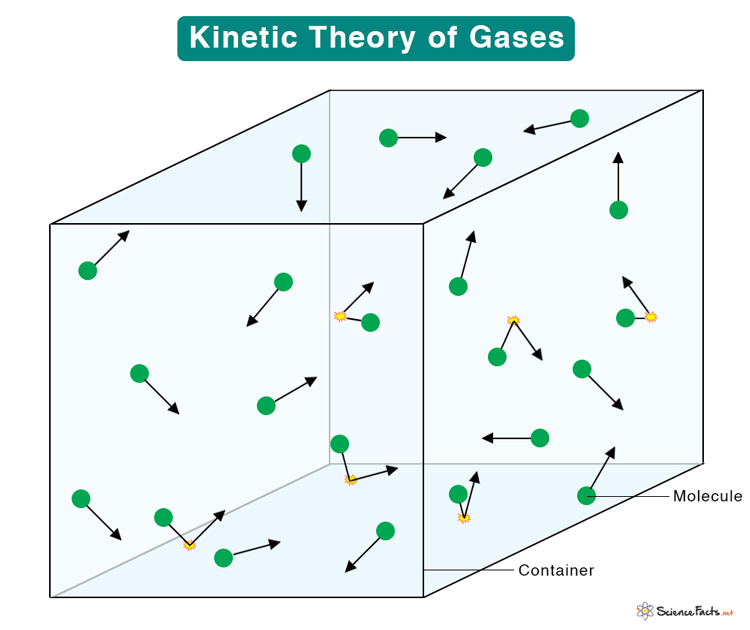
Image courtesy of Science Facts
Kinetic Theory and Gas Pressure:
- Microscopic Interpretation: At the microscopic level, gas pressure can be understood as the cumulative effect of numerous collisions of gas particles against the container walls.
- Macroscopic Correlations: This microscopic understanding explains the observable behaviours of gases under varying conditions in laboratories and natural settings.
Detailed Analysis of Gas Pressure and Particle Collisions
The relationship between gas pressure and particle collisions is intricate and forms a key concept in the kinetic theory.
Factors Influencing Gas Pressure:
- Particle Velocity: The speed of gas particles plays a critical role. Faster particles result in more frequent and forceful collisions, thus increasing the gas pressure.
- Container Dimensions: The size of the container directly affects the pressure. In smaller containers, particles collide more frequently with the walls, which increases the pressure.
- Number of Particles: An increase in the number of particles in a given volume escalates the collision frequency, thereby raising the pressure.
- Temperature Effects: As the temperature rises, particles move faster, leading to an increase in pressure due to more forceful collisions.
Practical Applications and Examples
The principles of gas pressure and particle collisions are not just theoretical concepts but have numerous practical applications.
Real-World Applications:
- Aerosol Sprays: The pressurised gas in aerosol cans exemplifies the principles of gas pressure and particle collisions.
- Automotive Airbags: The rapid generation of gas pressure is crucial in the functioning of airbags in vehicles.
- Weather Balloons: The expansion and contraction of gases in weather balloons demonstrate the effects of temperature on gas pressure.

Image courtesy of lilyl
Experimental Observations and Calculations
In a laboratory setting, the relationship between gas pressure and particle collisions can be observed and quantified through various experiments.
Laboratory Experiments:
- Pressure Measurement: Experiments involving the measurement of gas pressure under different temperatures and volumes can illustrate the principles discussed.
- Graphical Analysis: Plotting pressure against temperature or volume can provide a visual representation of the kinetic theory in action.
Understanding gas pressure in terms of particle collisions and the kinetic theory is pivotal in the field of chemistry. This knowledge is not only crucial for academic purposes but also has significant practical applications in various industries and everyday life, underscoring its importance in the A-level Chemistry curriculum.
In summary, this section has provided a comprehensive overview of gas pressure and particle collisions, grounded in the kinetic theory of gases. It has covered the basic concepts, detailed analysis, practical applications, and experimental observations, offering a thorough understanding for A-level Chemistry students.
FAQ
In deep-sea diving, the ambient pressure increases significantly with depth due to the weight of the overlying water. The pressure increases by approximately one atmosphere for every 10 meters of water depth. This increased pressure affects the behaviour of gases according to the principles of the kinetic theory. The gas in divers’ tanks and in their bodies must be adjusted to match the ambient pressure to avoid decompression sickness and other diving-related hazards. As pressure increases, gases become more soluble in liquids; this can lead to nitrogen dissolving in the diver's blood, which can cause decompression sickness if not managed correctly. Therefore, adjusting gas pressures is essential for safe diving to ensure that the gases in the body and equipment behave predictably and safely under increased pressure.
Meteorologists heavily rely on the concept of gas pressure for weather prediction. Atmospheric pressure is a key indicator of weather patterns. High-pressure systems generally bring clear, calm weather, while low-pressure systems are often associated with clouds and precipitation. The movement and interaction of different pressure systems are crucial in determining weather changes. Additionally, the pressure gradient, which is the rate of pressure change over a distance, is critical in predicting wind patterns. Winds generally move from areas of high pressure to low pressure. By analysing changes in atmospheric pressure, meteorologists can predict wind speeds and directions, which are essential for forecasting weather conditions, including storms and cyclones. Understanding gas pressure in the atmosphere is thus fundamental in meteorology and plays a vital role in weather forecasting and analysis.
Understanding gas pressure is crucial in chemical reactions involving gases, as it directly influences the rate and extent of the reaction. According to Le Chatelier’s principle, a change in pressure can shift the equilibrium position of a gaseous reaction. For reactions involving unequal numbers of moles of gaseous reactants and products, changes in pressure can significantly alter the yield of the reaction. Additionally, the rate of a gas-phase reaction is affected by the frequency of collisions between reactant molecules, which is influenced by gas pressure. Higher pressures increase the concentration of gas molecules, leading to more frequent collisions and potentially a faster reaction rate. Therefore, control and understanding of gas pressure are fundamental in optimising conditions for desired chemical reactions.
Humidity refers to the amount of water vapour present in the air. Water molecules, when added to the air, increase the total number of particles in the air. According to the kinetic theory, the pressure of a gas is proportional to the number of particles in a given volume. As humidity increases, more water vapour molecules are present, which increases the number of collisions between gas particles and the walls of the container (in this case, the atmosphere). Consequently, higher humidity results in a slight increase in air pressure. However, this effect is often subtle compared to other factors affecting atmospheric pressure, such as temperature and altitude.
Gas pressure varies significantly with altitude due to changes in the density of air. At higher altitudes, the atmosphere becomes less dense, meaning there are fewer air molecules in a given volume. According to the kinetic theory, gas pressure is a result of collisions of particles with the walls of their container. At higher altitudes, as the number of air molecules decreases, the frequency of these collisions also decreases, leading to a reduction in air pressure. This is why mountaintops have significantly lower air pressure compared to sea level. Additionally, gravitational forces are weaker at higher altitudes, further contributing to the reduction in air pressure. This variation in pressure is a critical consideration in fields like aviation and mountaineering, where adjustments need to be made for lower atmospheric pressure.
Practice Questions
An excellent A-level Chemistry student's answer:
The kinetic theory of gases posits that gas particles are in constant, random motion and that their collisions with container walls result in gas pressure. As temperature increases, the kinetic energy of the gas particles also increases. This elevation in kinetic energy translates into a higher velocity of the gas particles. Consequently, with faster-moving particles, the frequency and force of collisions against the container walls escalate. This increase in collision rate and force heightens the gas pressure. Therefore, according to the kinetic theory, an increase in temperature leads to an increase in gas pressure due to the more energetic and frequent particle collisions.
An excellent A-level Chemistry student's answer:
According to the kinetic theory of gases, gas pressure is created by particles colliding with the walls of their container. When the size of the gas container is reduced, the available space for the gas particles to move diminishes. Consequently, this leads to an increased frequency of particle collisions with the container walls, as the same number of particles is confined to a smaller volume. More frequent collisions result in a higher exerted pressure on the walls of the container. Therefore, reducing the size of the container increases the pressure of the gas inside, as per the kinetic theory of gases.

