In-depth examination of experimental methodologies to study the impact of temperature and substrate concentration on yeast respiration, focusing on the use of redox indicators for accurate and reliable results.
Introduction to Respiration Experiments
Investigating yeast respiration provides insights into fundamental biological processes. This exploration is crucial for understanding how external factors like temperature and substrate concentration can influence cellular respiration, a process essential for life.
The Role of Redox Indicators in Respiration Studies
Redox indicators are pivotal in visually tracking the respiration process in yeast, revealing changes in the cellular oxidation-reduction state.
Selection and Functionality of Redox Indicators
- Methylene Blue: A widely chosen indicator due to its distinct colour shift from blue (oxidised) to colourless (reduced) under active respiration.
- Resazurin: Offers a nuanced colour transition from blue to pink and then colourless, enabling finer observation of the respiration process.
Image courtesy of Vinicius Waldow
Application in Yeast Respiration
- Mix redox indicators with yeast cultures in solution.
- Observe and record the time taken for the solution to change colour, indicating respiration activity.
Examining Temperature Effects on Yeast Respiration
The rate of biochemical reactions in yeast, including respiration, is temperature-dependent.
Experiment Design for Temperature Variance
- Selecting Temperature Ranges: Experiments should include a broad temperature range, typically from 10°C to 40°C.
- Maintaining Controlled Conditions: Use accurate temperature control methods, like water baths, to ensure consistent experimental conditions.
- Data Recording: Carefully time the colour change in the redox indicator at each temperature and document results systematically.
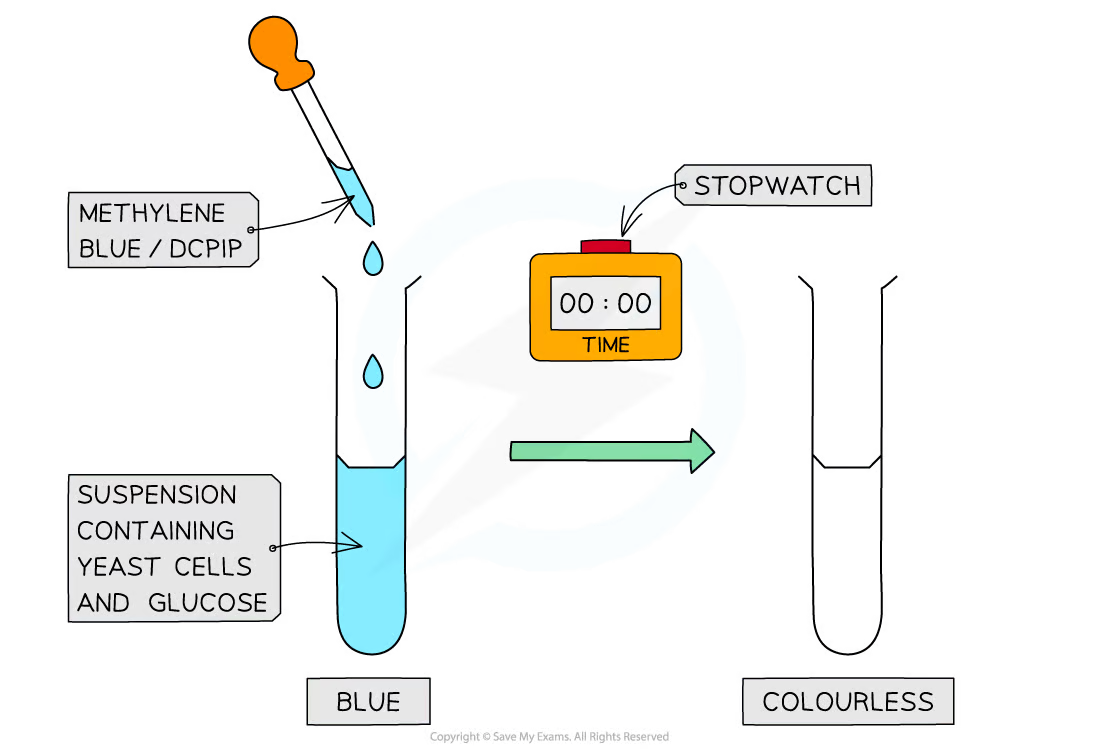
Image courtesy of Save My Exams
Interpreting Temperature Experiment Data
- Expect increased respiration rates with rising temperatures up to a species-specific optimum.
- Analyse how enzyme denaturation at high temperatures affects respiration.
Investigating Substrate Concentration Influence
Substrate concentration, especially glucose, is a critical factor in yeast respiration rate.
Setting Up Substrate Concentration Experiments
- Gradient of Concentrations: Prepare a series of yeast solutions with increasing glucose concentrations.
- Maintaining Consistent Other Variables: Ensure that temperature and pH are constant across all test samples.
- Observing Respiration Rates: Track the time for colour changes in the redox indicator as a measure of respiration rate.
Analysing Substrate Concentration Results
- Identify the concentration at which the respiration rate plateaus, indicating enzyme saturation.
- Compare respiration rates at different concentrations to understand substrate affinity.
IB Biology Tutor Tip: Understanding the optimum conditions for yeast respiration, such as ideal temperature and substrate concentration, is crucial for designing experiments that accurately reflect cellular processes.
Implementing Control Measures for Accurate Results
Accurate and reliable results are fundamental in scientific experiments.
Repetition for Consistency
- Perform each experiment several times to check for consistency in the results.
Standardising Experimental Conditions
- Use the same yeast strain and batch of reagents to eliminate variability.
- Calibrate measuring instruments regularly to ensure precision.
Controlling Environmental Factors
- Conduct experiments in a stable environment to reduce external influences like light and air currents.
Best Practices in Experimental Design
A meticulously planned experiment is crucial for meaningful data.
Formulating Hypotheses
- Develop specific hypotheses regarding the impact of temperature and substrate concentration on yeast respiration.
Detailed Methodological Planning
- Clearly outline each step, from preparing yeast cultures to measuring and recording data.
Systematic Data Collection and Analysis
- Collect data in an organised manner, using standardised units and methods.
- Apply statistical analysis techniques to interpret the data accurately.
Rigorous Documentation
- Maintain detailed records of experimental procedures, observations, and findings for future reference and validation.
Advanced Techniques in Respirometry
Beyond basic redox indicators, respirometry offers a more precise way to measure respiration rates.
Respirometer Setup
- A respirometer can precisely measure oxygen consumption or carbon dioxide production in yeast cultures.
- Set up the respirometer with a sealed chamber containing yeast culture and a device to measure gas exchange.
Interpreting Respirometer Data
- Analyse the data to determine the rate of oxygen consumption or carbon dioxide production, offering insight into the metabolic rate of the yeast under different conditions.
Experimental Variables and Their Control
Understanding and controlling experimental variables is key to obtaining valid results.
Temperature as a Variable
- Monitor and adjust temperature accurately, considering its profound impact on enzymatic activity.
Substrate Concentration Variability
- Prepare substrate solutions with meticulous accuracy to ensure consistent concentration levels.
pH and Its Role
- Maintain a constant pH in the yeast culture, as pH variations can significantly affect enzyme activity and cell health.
IB Tutor Advice: When revising, focus on the relationship between enzyme activity and temperature/substrate concentration, and how this affects yeast respiration rates, to predict outcomes of similar biological processes in exam questions.
Conclusion
Studying the effects of temperature and substrate concentration on yeast respiration through methodical experimentation and the use of redox indicators or respirometry is crucial in understanding cellular processes. Adhering to strict experimental protocols ensures the accuracy and reliability of the findings, contributing significantly to our comprehension of biological systems.
FAQ
Yes, redox indicators can be used to study anaerobic respiration in yeast. Anaerobic respiration, or fermentation in yeast, involves the conversion of glucose to ethanol and carbon dioxide in the absence of oxygen. Redox indicators like methylene blue can be employed to observe changes in the redox state of the cell during anaerobic respiration. In the absence of oxygen, methylene blue remains in its reduced form, indicating anaerobic conditions. By comparing the rate of colour change or the duration for which the colour remains unchanged in both aerobic and anaerobic conditions, one can draw conclusions about the efficiency and characteristics of anaerobic respiration in yeast.
Oxygen plays a crucial role in yeast respiration experiments, especially when using redox indicators. In aerobic respiration, oxygen is the final electron acceptor in the electron transport chain, and its presence is essential for the complete oxidation of glucose to carbon dioxide and water. In experiments using redox indicators, the availability of oxygen can affect the rate of respiration and, consequently, the rate of colour change in the indicator. To monitor oxygen levels, respirometers or oxygen sensors can be used. These devices measure the concentration or pressure of oxygen in the experimental setup, providing data on how oxygen availability influences the respiration rate. Additionally, by controlling oxygen levels, researchers can study both aerobic and anaerobic respiration processes in yeast.
Besides temperature and substrate concentration, several other factors can influence yeast respiration, including pH levels, oxygen availability, and the presence of inhibitors or activators.
- pH Levels: The pH of the environment can affect enzyme activity in yeast cells. This can be investigated by conducting experiments where the pH is systematically varied and observing the impact on respiration rates using redox indicators or respirometry.
- Oxygen Availability: Oxygen is crucial for aerobic respiration. Its influence can be studied by comparing respiration rates in aerobic and anaerobic conditions using a respirometer or observing colour changes in redox indicators.
- Presence of Inhibitors or Activators: Certain chemicals can inhibit or enhance enzyme activity in the respiration pathway. Investigating the impact of such substances involves adding them to the yeast culture and monitoring the changes in respiration rate, which can be done using respirometry or redox indicators.
These factors can be independently manipulated in controlled experiments to understand their specific effects on yeast respiration.
A change in pH can significantly affect yeast respiration. Yeast cells contain enzymes that catalyse the various steps of respiration, and these enzymes have optimal pH ranges within which they function most effectively. Deviations from this optimal pH can lead to a decrease in enzyme activity, thereby slowing down the respiration process. For instance, acidic or highly alkaline conditions may denature the enzymes or alter their active sites, reducing the rate of respiration. This effect can be studied using redox indicators by setting up experiments where the pH of the yeast culture is systematically varied. The redox indicator would show variations in the rate of colour change, indicating changes in the respiration rate under different pH conditions. By correlating the pH levels with the speed of the redox indicator’s colour change, one can infer the impact of pH on yeast respiration.
Methylene blue is preferred in yeast respiration experiments for several reasons. Firstly, its distinct colour change from blue to colourless in reduced form makes it easy to observe and interpret the results, especially for determining the endpoint of the experiment. This clear change is due to its ability to accept electrons during the reduction process in respiration, making it an effective marker of cellular activity. Secondly, methylene blue is non-toxic to yeast cells at low concentrations, ensuring that it does not interfere with the respiration process being studied. Its compatibility with a wide range of experimental conditions, including various temperatures and pH levels, adds to its suitability. Finally, the simplicity of using methylene blue, combined with its cost-effectiveness, makes it an accessible tool for educational and research laboratories.
Practice Questions
In yeast respiration, as the temperature increases, the rate of respiration initially rises due to increased kinetic energy, enhancing enzyme-substrate collisions. This continues until the optimal temperature is reached, beyond which enzymes begin to denature, reducing the respiration rate. Experimentally, this can be observed using a redox indicator like methylene blue, which changes colour when reduced. By measuring the time taken for the colour change at various temperatures, one can graphically represent the relationship between temperature and respiration rate. The steepest slope indicates the highest respiration rate, typically at the optimum temperature.
Substrate concentration affects yeast respiration rate in a manner described by the Michaelis-Menten kinetics. Initially, an increase in substrate (e.g., glucose) concentration leads to a corresponding increase in respiration rate due to more frequent collisions between substrate molecules and active sites of enzymes. However, beyond a certain concentration, the rate plateaus as enzyme active sites become saturated, and further increases in substrate concentration do not enhance the rate. To investigate this, one could set up an experiment with varying concentrations of glucose, using a redox indicator to observe the rate of respiration at each concentration, and then plotting these rates against the concentrations.

