Understanding the partition coefficient (Kpc) is fundamental in the field of chemistry, especially when dealing with the distribution of a solute between two immiscible solvents. This section comprehensively explores the various factors that influence the numerical value of Kpc, emphasizing the role of solute and solvent polarities and their implications in different solvent systems.
Introduction to Partition Coefficients
Partition coefficients (Kpc) are pivotal in predicting how substances distribute between two distinct phases, typically an organic solvent and water. The concept of Kpc provides insight into the solubility behaviour of compounds and is essential in various chemical and pharmaceutical applications. Accurate knowledge of these coefficients aids in understanding the transportation and bioavailability of drugs, the behaviour of chemicals in the environment, and the principles of extraction processes.
Understanding Polarity in Solvents and Solutes
The Concept of Polarity
- Definition and Importance: Polarity in chemistry refers to the uneven distribution of electrons across a molecule, resulting in regions of partial positive and negative charges. This phenomenon significantly influences how molecules interact with one another, especially in terms of solubility and reactivity.
- Polar Molecules: These are molecules with a significant difference in electronegativity between bonded atoms, leading to a dipole moment. Water is a classic example, with its oxygen atom having a partial negative charge and hydrogen atoms a partial positive charge.
- Non-Polar Molecules: Molecules like methane or oils, where the electronegativity difference between the atoms is minimal, resulting in a fairly even distribution of electrical charge.
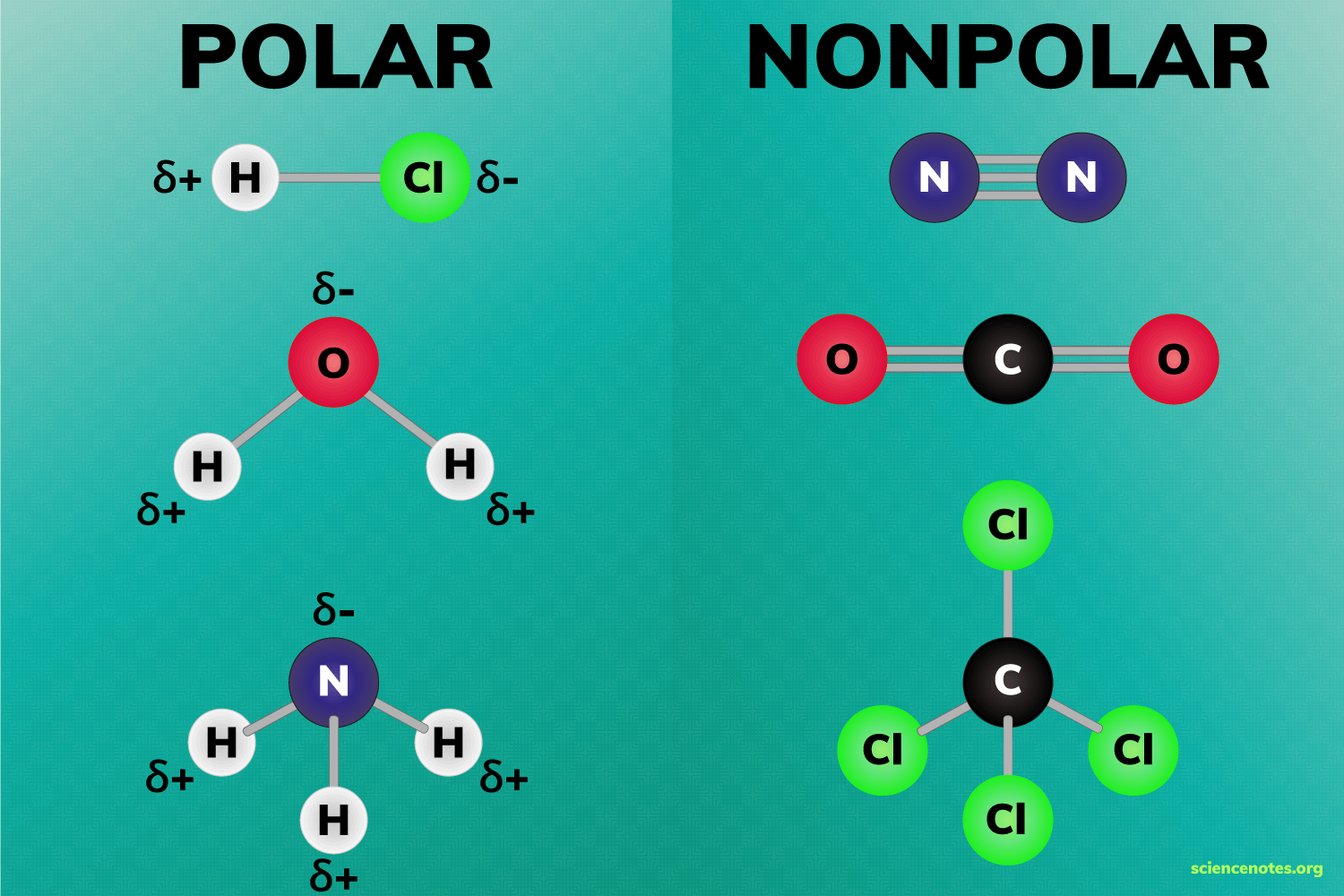
Image courtesy of Science Notes
Impact of Polarity on Solubility
- Polar Solvents: These solvents, such as water, are efficient at dissolving ionic compounds and other polar substances due to the ability to form dipole-dipole interactions or hydrogen bonds.
- Non-Polar Solvents: Solvents like hexane dissolve non-polar substances better. This is because non-polar molecules are held together by relatively weaker van der Waals forces, which are easily disrupted by similar non-polar solvents.
Factors Influencing Kpc
Polarity and Its Effects
- Interaction Between Solute and Solvent: The solubility of a compound in a solvent and hence its partition coefficient is profoundly influenced by the polarity of both. For instance, polar solutes tend to have higher solubility in polar solvents, altering the Kpc.
- Polar vs Non-Polar Systems: A polar solute like sodium chloride will have a significantly different partition coefficient in water (polar) compared to an organic solvent like benzene (non-polar).
Molecular Size and Structure
- Molecular Size: Generally, larger molecules have lower solubility, which can affect the Kpc. For example, large organic molecules might be more soluble in organic solvents than in water.
- Structural Factors: The shape of a molecule and the presence of various functional groups greatly influence its interaction with different solvents. For instance, molecules with large non-polar regions tend to be more soluble in non-polar solvents.

Image courtesy of zaie
Temperature
- Solubility Dependence: The solubility of a compound in a solvent generally increases with temperature. However, this trend can vary depending on the nature of the solute and solvent.
- Temperature and Kpc: Changes in temperature can lead to significant variations in Kpc values. For a given solute, an increase in temperature might increase its solubility in one solvent more than the other, altering the partition coefficient.

Image courtesy of upklyak
pH of the Solution
- Ionization and Solubility: In aqueous solutions, the pH can affect the ionization state of solutes, particularly for acidic or basic compounds. This change in ionization can significantly influence solubility and, consequently, the partition coefficient.
- pH-Dependent Partitioning: For weak acids and bases, the partition coefficient can vary dramatically with pH. This is crucial in pharmaceuticals, where the drug's efficacy can depend on its ability to cross biological membranes, which in turn is influenced by its ionization state at different pH levels.
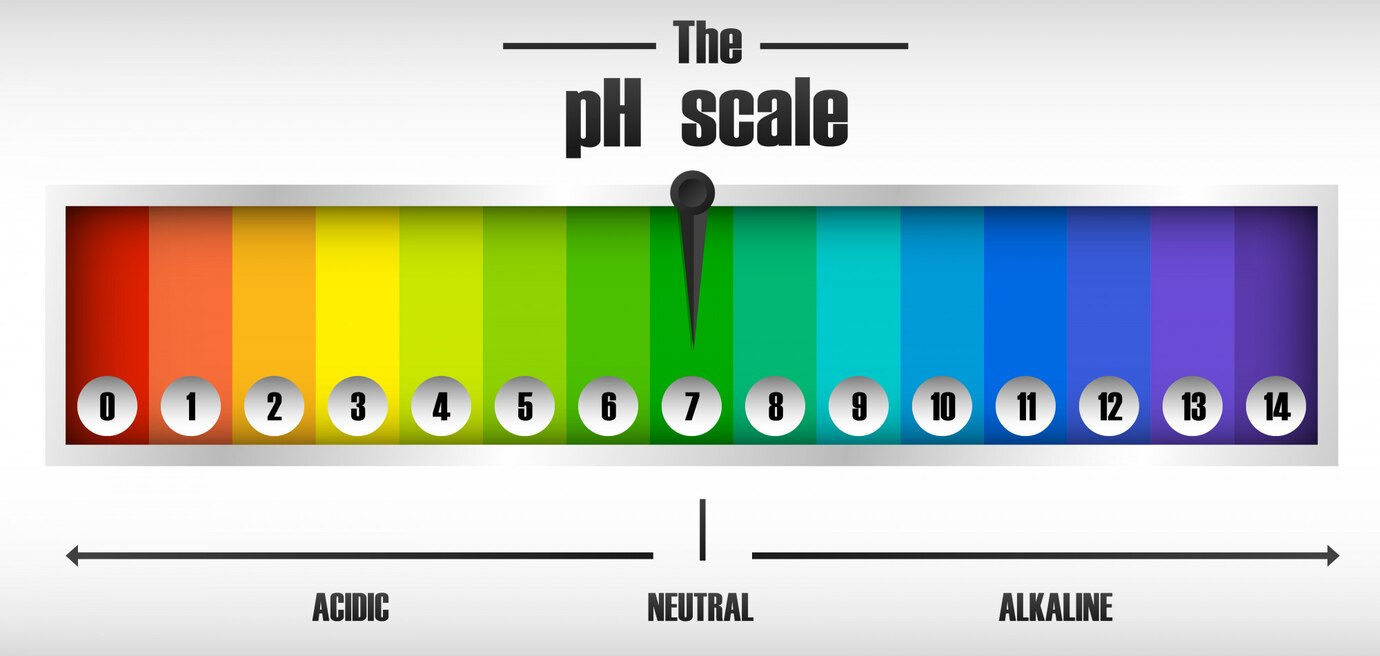
Image courtesy of brgfx on freepik
Applications and Implications
In Drug Design and Pharmacokinetics
- Predicting Drug Behaviour: The Kpc of a drug helps predict its absorption, distribution, metabolism, and excretion in the body. For instance, a drug needs to have a balanced partition coefficient to be both water-soluble (for blood transport) and lipid-soluble (to cross cell membranes).
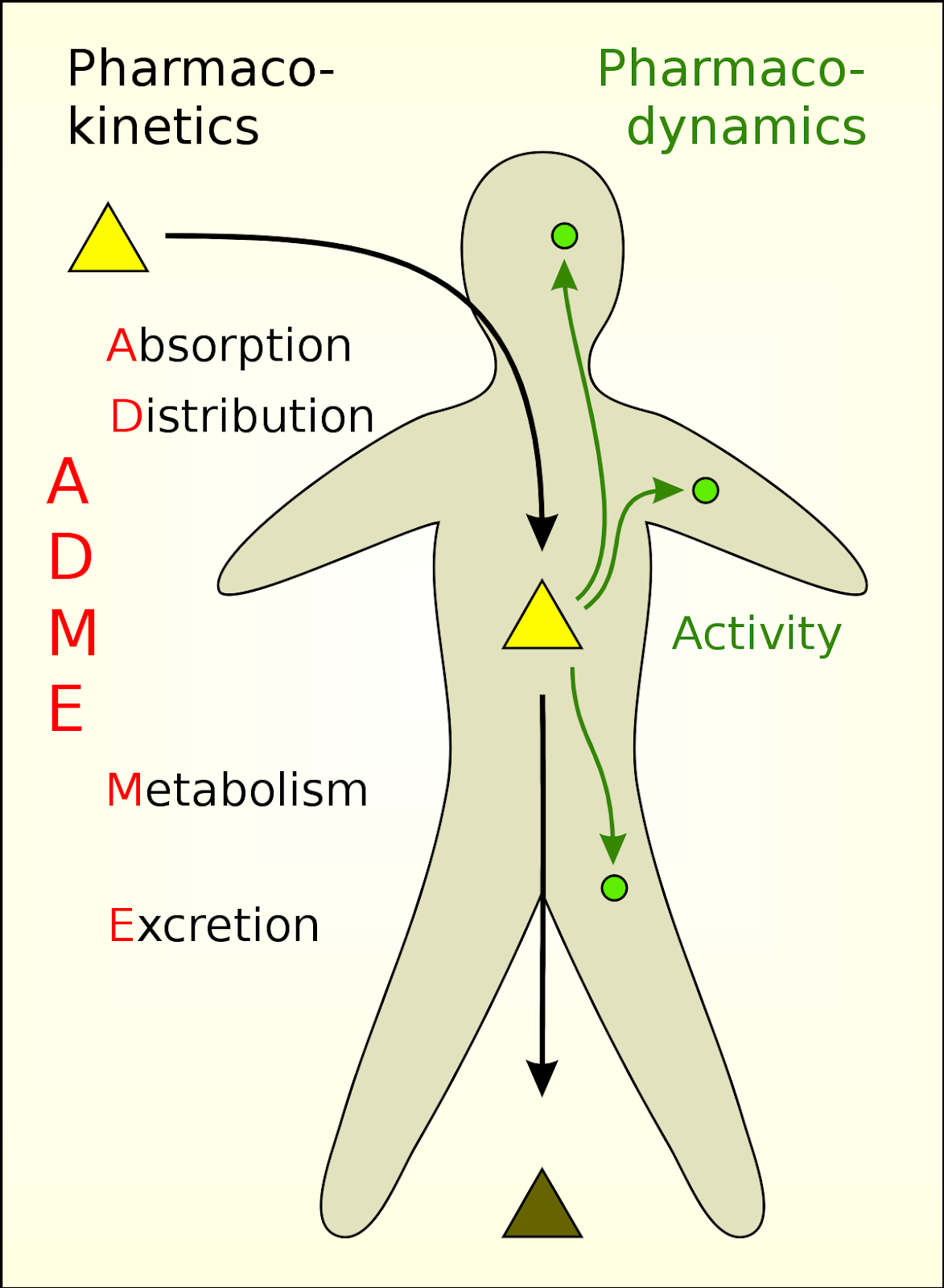
Image courtesy of Scivit
In Environmental Chemistry
- Pollutant Distribution: Understanding the partition coefficients of pollutants helps in predicting their behaviour in the environment. For instance, chemicals with a high partition coefficient for fat are likely to bioaccumulate in organisms.
In Analytical Chemistry
- Use in Chromatography: Partition coefficients are fundamental in chromatography, where they influence the separation of components in a mixture based on their distribution between the stationary and mobile phases.
Case Studies and Examples
Example 1: Aspirin in Water and Oil
- This example illustrates how aspirin's weak acidic nature influences its Kpc between water (polar) and oil (non-polar). The ionization of aspirin in water reduces its solubility in the oil phase, affecting the partition coefficient.

Image courtesy of Racool_studio on freepik
Example 2: Dye Distribution
- The distribution of a dye between water and an organic solvent can be analyzed to understand the impact of molecular structure and polarity on its Kpc. Dyes with polar functional groups might preferentially dissolve in water, while non-polar dyes may have a higher affinity for organic solvents.
Practical Applications in the Laboratory
Experimentation with Partition Coefficients
- Laboratory Experiments: Students can perform simple experiments to observe the effects of polarity, temperature, and pH on the partition coefficient of a given solute. These experiments can involve measuring the concentration of a solute in two immiscible solvents under varying conditions.
FAQ
The molecular symmetry of a solute can have a significant impact on its partition coefficient. Symmetrical molecules tend to be non-polar or have balanced polarity, as the symmetry leads to an even distribution of electrical charge across the molecule. This uniform charge distribution makes symmetrical molecules more soluble in non-polar solvents and less soluble in polar solvents like water. As a result, symmetrical molecules generally have higher partition coefficients in systems involving non-polar and polar solvent combinations, indicating a preference for the non-polar phase. Conversely, asymmetrical molecules often have uneven charge distributions, resulting in greater polarity. This increased polarity enhances their solubility in polar solvents, thereby lowering the partition coefficient in a polar-non-polar solvent system. It's important to consider that while symmetry influences solubility and partition coefficients, other factors like molecular size, functional groups, and specific interactions with the solvent molecules (like hydrogen bonding or dipole interactions) also play critical roles in determining the overall partitioning behaviour of a solute.
The ionic strength of a solution can significantly influence the partition coefficient of ionic solutes. Ionic strength refers to a measure of the concentration of ions in a solution and their ability to affect the behaviour of other ions in the solution. In the context of partition coefficients, an increase in ionic strength in the aqueous phase can lead to a phenomenon known as "salting out." This occurs because the presence of additional ions in the solution competes with the ionic solute for solvation by water molecules. As a result, the solubility of the ionic solute in the aqueous phase decreases, causing an increase in its partition coefficient. This means the ionic solute becomes more likely to partition into the non-polar phase. The "salting out" effect is particularly noticeable with ionic compounds that are not strongly solvated or are only moderately polar. In practical applications, manipulating the ionic strength of a solution is a common technique used to control the distribution of ionic solutes between phases, such as in extraction processes or in adjusting the properties of a solution for analytical or industrial purposes.
The presence of chiral centres in a molecule can affect its partition coefficient, but the impact is usually subtle and dependent on the specific interactions of the chiral molecule with the solvents. Chirality refers to a molecular property where a molecule cannot be superimposed on its mirror image, much like left and right hands. Chiral molecules often show different behaviours in biological systems due to their ability to interact differently with other chiral environments, such as enzymes or receptors. However, when considering partition coefficients, which are primarily influenced by physical properties like solubility, the effect of chirality is less pronounced unless the solvents themselves exhibit chiral properties or the partitioning involves chiral recognition mechanisms. In achiral environments, the solubility and therefore the partition coefficients of enantiomers (chiral mirror-image molecules) are generally similar. However, in cases where chiral interactions come into play, such as in chiral chromatography or enantioselective synthesis, the partitioning behaviour can differ significantly between enantiomers. Thus, while chirality can affect partition coefficients under certain conditions, the extent of this effect is highly dependent on the specific chemical system and the presence of chiral interactions.
Yes, the partition coefficient can be a valuable predictor of a chemical's bioaccumulation potential in living organisms. Bioaccumulation refers to the process where a chemical substance accumulates in an organism at a rate faster than it is metabolised or excreted. Chemicals with high partition coefficients, particularly those favouring lipid (fat) solvents, tend to accumulate in the fatty tissues of organisms. This is because organisms, especially animals, have significant fat content, and substances that are more soluble in fats than in water (i.e., have a high partition coefficient) will preferentially dissolve in and remain within these fatty tissues. For example, many persistent organic pollutants (POPs) have high partition coefficients, which contributes to their ability to bioaccumulate and even biomagnify in food chains. It's important to note that while a high partition coefficient is an indicator of potential bioaccumulation, other factors such as the chemical's metabolic stability, its ability to cross biological membranes, and its environmental persistence also play crucial roles in determining its actual bioaccumulation in a specific organism or ecosystem.
The presence of hydrogen bonding in a solute significantly impacts its partition coefficient when distributed between water (a polar solvent) and a non-polar solvent. Hydrogen bonds are strong dipole-dipole interactions occurring in molecules where hydrogen is bonded to highly electronegative atoms like oxygen, nitrogen, or fluorine. Solute molecules capable of hydrogen bonding interact more strongly with water due to its polar nature and the ability to form hydrogen bonds. This interaction increases the solute's solubility in water, resulting in a lower partition coefficient, as the solute prefers the aqueous phase over the non-polar phase. On the other hand, in a non-polar solvent, where hydrogen bonding is not favourable, the solute's solubility decreases. Therefore, the solute's affinity for water is significantly enhanced by hydrogen bonding, leading to a distribution that favours the aqueous phase over the non-polar phase. This effect is particularly noticeable with solutes that have multiple sites for hydrogen bonding, as they can form extensive hydrogen-bonding networks with water molecules, drastically altering the partition behaviour.
Practice Questions
The partition coefficient of a weak acid, like acetic acid, varies significantly with the pH of the water. In a more acidic environment (lower pH), the weak acid remains largely undissociated, favouring its solubility in the organic phase. This results in a higher partition coefficient. Conversely, at higher pH levels, the weak acid ionises to a greater extent, becoming more soluble in the aqueous phase and less in the organic phase, thus lowering the partition coefficient. An excellent answer would demonstrate understanding that the partition coefficient is not a fixed value but varies with environmental conditions such as pH, impacting the distribution of the solute between the two phases.
The partition coefficient of a non-polar solute, such as benzene, between water and an organic solvent, like hexane, is influenced by temperature. Generally, an increase in temperature enhances the solubility of non-polar solutes in both polar and non-polar solvents. However, the solubility tends to increase more in the non-polar solvent (hexane) than in water. This results in a higher partition coefficient at elevated temperatures, indicating a greater preference of the solute for the organic phase. A thorough answer would also note that the extent of this effect can vary depending on the specific properties of the solute and solvents involved.

