Enthalpy changes play a pivotal role in chemical thermodynamics, especially in processes involving hydration and solution. This comprehensive guide is tailored for A-level Chemistry students, aiming to deepen understanding of the concepts of ΔH_hydration and ΔH_solution. It covers their definitions, the methodologies for their calculation, and the intricate energy changes that occur when ions dissolve in water.
Introduction to Enthalpy Changes
Understanding enthalpy changes is crucial in the realm of chemical reactions and processes. Enthalpy (H) is a measure of the total heat content of a system under constant pressure. Changes in enthalpy (ΔH) provide insights into the energy exchanged between the system and its surroundings during chemical reactions.
Enthalpy Change of Hydration (ΔH_hydration)
Definition and Significance
- Definition: ΔH_hydration is defined as the enthalpy change when one mole of gaseous ions is completely hydrated by water molecules.
- Significance: It is a key concept in understanding how ionic substances interact with water, an essential solvent in chemistry.
Energy Considerations
- Exothermic Nature: The hydration of ions is typically exothermic. As ions get surrounded by water molecules, energy is released due to the formation of ion-dipole interactions.
- Magnitude of ΔH_hydration: The magnitude depends on the ion's charge density; higher charge densities typically result in larger exothermic values.
Enthalpy Change of Solution (ΔH_solution)
Definition and Importance
- Definition: ΔH_solution is the enthalpy change when one mole of a solute dissolves in sufficient solvent to form an ideal solution.
- Importance: It helps in understanding the energetics of solution formation, which is fundamental in many chemical and biological processes.
Energy Dynamics in Solution Formation
- Process Overview: In dissolving an ionic compound, the lattice must first break apart (endothermic) and then ions must become hydrated (exothermic).
- Net Enthalpy Change: ΔH_solution is the result of these competing processes and can be either endothermic or exothermic, depending on the substance.
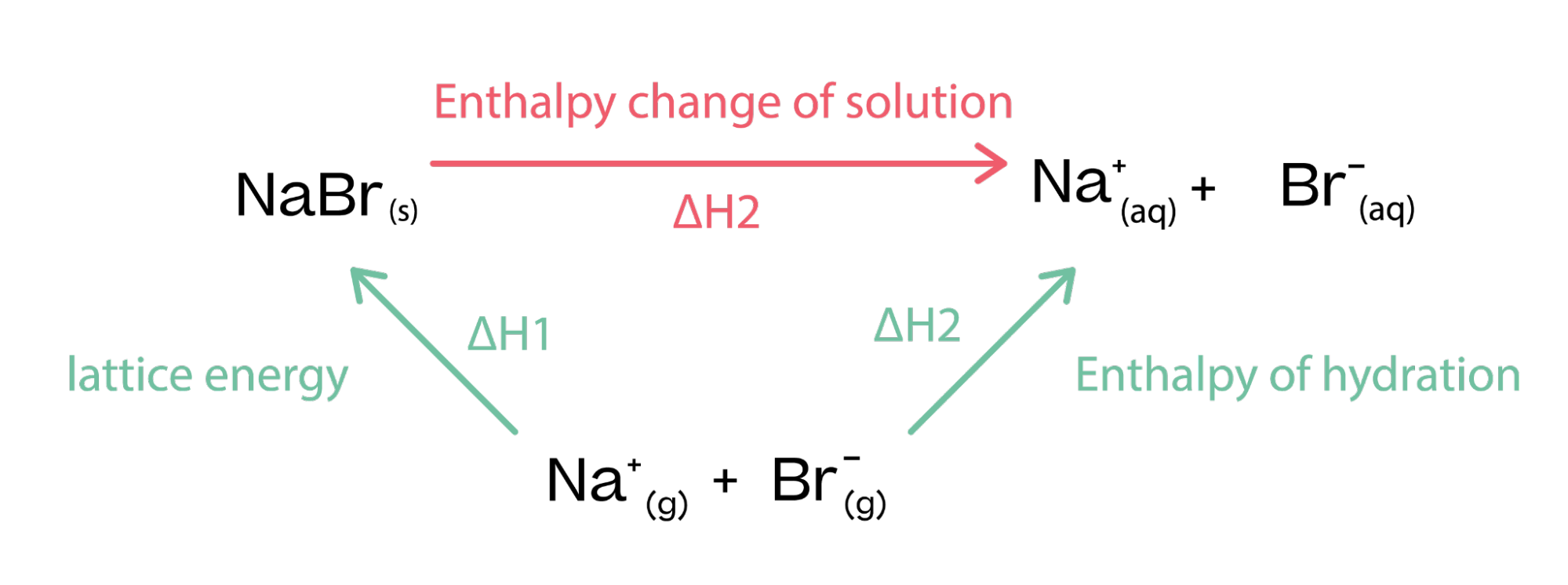
Image courtesy of evulpo
Calculations of ΔH_hydration and ΔH_solution
Calculating ΔH_hydration
- Experimental Methods: Typically determined using calorimetric techniques, where the heat change upon dissolving a known amount of the ion in water is measured.
- Hess's Law Application: Involves using known enthalpies of other reactions to calculate ΔH_hydration indirectly.
Calculating ΔH_solution
- Calorimetry in Practice: The heat change is measured when a solute dissolves in a solvent. The experiment must be carefully controlled to ensure accuracy.
- Theoretical Calculations: Hess's law again plays a crucial role. It involves using the enthalpy changes of formation, lattice energy, and hydration to compute ΔH_solution.
Energy Changes in Ionic Solutions
Dissolution Process
- Ionic Bonds and Water Interaction: The dissolution of an ionic compound in water is a two-step process. First, the ionic lattice breaks down (endothermic), and then ions are hydrated by water molecules (exothermic).
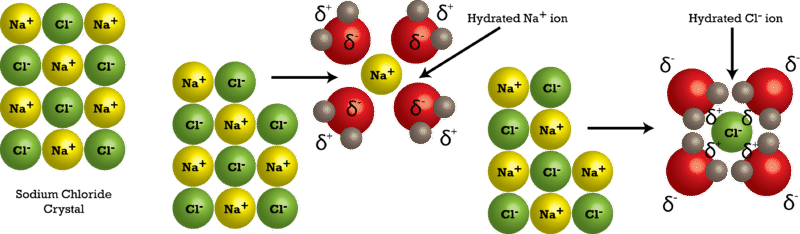
Image courtesy of CK-12
Hydration Energy
- Understanding Hydration Energy: This concept refers to the energy change when ions form ion-dipole bonds with water molecules.
- Factors Affecting Hydration Energy: The size (radius) and charge of the ion significantly influence the hydration energy. Smaller ions with higher charges have greater hydration energies.
Energy Profile of Dissolving Ions
- Lattice Energy Consideration: The energy required to break the ionic lattice is a key factor in determining the overall energy change.
- Net Enthalpy Change (ΔH_solution): The sum of lattice energy and hydration energy gives the net enthalpy change. The balance between these energies determines the endothermic or exothermic nature of the solution process.
Practical Applications and Examples
Laboratory Demonstrations
- Conducting Experiments: Students can measure ΔH_solution for various salts to understand the concept practically.
- Data Interpretation: Analyzing the results to relate the theoretical concepts with the experimental findings.
Real-World Examples
- Industrial Relevance: These concepts are pivotal in industries like mineral processing, where the solubility of compounds is key.
- Environmental Chemistry: The dissolving of salts in natural water bodies can affect the environmental balance, making the understanding of these processes crucial.
Summary and Key Takeaways
- Fundamental Concepts: ΔH_hydration and ΔH_solution are essential in understanding the energetics of ionic solutions.
- Experimental and Theoretical Methods: Both experimental (calorimetry) and theoretical (Hess's law) methods are used to calculate these enthalpy changes.
- Energy Balance: The dissolution of ionic compounds involves a balance of endothermic and exothermic processes.
- Real-World Applications: These concepts find applications in various fields, highlighting their importance beyond the classroom.
Through this detailed exploration of enthalpy changes in hydration and solution, A-level chemistry students gain a profound understanding of these essential thermodynamic concepts. The knowledge not only supports academic pursuits but also provides a foundation for real-world applications in various scientific and industrial fields.
FAQ
Different ions have varying enthalpies of hydration (ΔH_hydration) primarily due to differences in their charge and size (ionic radius). The enthalpy of hydration is more exothermic for ions with higher charges and smaller ionic radii. This is because ions with a higher charge density (charge-to-radius ratio) have stronger electrostatic attractions to the dipole of the water molecules. For instance, a smaller ion with a higher charge will pull the dipole water molecules closer and more strongly than a larger ion with the same charge, leading to a greater release of energy during hydration. This principle is why, for example, a lithium ion (Li⁺), which is small and highly charged, has a more exothermic ΔH_hydration compared to a larger ion like potassium (K⁺), even though both are monovalent cations.
The polarity of the solvent significantly influences the enthalpy change of solution, ΔH_solution. In polar solvents like water, ionic compounds tend to dissolve more readily due to strong ion-dipole interactions. These interactions are crucial in stabilising the ions in the solvent, leading to a more exothermic ΔH_solution. For example, in the case of common salts like sodium chloride, the positive end of the water molecule (hydrogen) attracts the chloride ion, while the negative end (oxygen) attracts the sodium ion. This strong interaction helps overcome the lattice energy of the salt, making the dissolution process energetically favourable. On the other hand, non-polar solvents, lacking this strong ion-dipole interaction, result in a less exothermic or even endothermic ΔH_solution for the same ionic compound. This is because, in non-polar solvents, the energy required to break the ionic lattice is not sufficiently compensated by the energy released through solvation, leading to a higher or even positive ΔH_solution value.
The enthalpy change of solution, ΔH_solution, can theoretically be zero, although this is rare in practice. This occurs when the energy required to break the ionic lattice of the solute (lattice energy) is exactly equal to the energy released when the solute ions are hydrated by the solvent molecules (hydration energy). In such a scenario, the energy absorbed in breaking the ionic bonds is perfectly balanced by the energy released in forming ion-dipole interactions, resulting in no net heat exchange with the surroundings. This condition is most likely to be approached when the solvent's ability to stabilise the ions is just sufficient to compensate for the lattice energy of the solute. However, it is essential to note that such perfect balance is rare due to the complexity of interactions involved in real systems. Factors like ion size, charge, solvent polarity, and temperature can all influence the exact values of lattice and hydration energies.
(ΔH_solution)?
Temperature plays a significant role in affecting the enthalpy change of solution (ΔH_solution). Generally, an increase in temperature leads to an increase in the solubility of most solid solutes in a liquid solvent. This is because the solubility of a solute is a dynamic equilibrium condition where the rate of dissolution and the rate of crystallisation are equal. Increasing the temperature typically increases the kinetic energy of the molecules, leading to a higher rate of dissolution. However, this does not always correlate with a more exothermic ΔH_solution. In some cases, higher temperatures can increase the solubility of a solute but also make the dissolution process more endothermic (less exothermic). This is because the increased kinetic energy can facilitate the overcoming of lattice energy, which might require more energy input. The exact effect of temperature on ΔH_solution is thus dependent on the specific intermolecular forces at play in the solute-solvent system.
The enthalpy change of solution (ΔH_solution) for a given solute can vary under different conditions. Several factors can cause these variations:
- Concentration of the Solution: At different concentrations, the interactions between the solute and solvent molecules can vary, leading to differences in ΔH_solution.
- Temperature: The solubility of substances generally increases with temperature, affecting the ΔH_solution. As temperature changes, it can alter the balance between the endothermic and exothermic processes involved in dissolution.
- Nature of the Solvent: Different solvents have varying abilities to solvate ions. A solvent's polarity and its own enthalpy of vaporisation can influence the enthalpy change of solution.
- Physical State of Solute: The physical form of the solute, such as whether it is crystalline or amorphous, can affect ΔH_solution. Different crystal structures have varying lattice energies.
- Pressure: Though less significant for solutions than for gases, changes in pressure can slightly affect the solubility and thus the ΔH_solution.
These variations highlight the importance of specifying conditions when discussing ΔH_solution values.
Practice Questions
The dissolution of sodium chloride in water is exothermic due to the net energy release when ionic bonds in the solid lattice are broken and new interactions are formed between the ions and water molecules. Initially, energy is absorbed to overcome the lattice energy of NaCl, breaking the ionic bonds (endothermic). However, when Na⁺ and Cl⁻ ions interact with water, ion-dipole forces are formed, releasing more energy than was absorbed (exothermic). The hydration of Na⁺ and Cl⁻ ions involves strong attractions between the ions and the polar water molecules, leading to a significant release of energy. The overall process is exothermic because the energy released during hydration outweighs the energy absorbed in breaking the lattice, as reflected in the negative ΔH_solution value of -3.7 kJ/mol.
The enthalpy change of hydration for Mg²⁺ is more exothermic compared to that for Na⁺. This difference is primarily due to the higher charge and smaller ionic radius of Mg²⁺ compared to Na⁺. Mg²⁺ has a charge of +2, while Na⁺ has a charge of +1. The higher charge density of Mg²⁺ leads to stronger ion-dipole interactions with water molecules, resulting in a greater release of energy during hydration. Additionally, the smaller ionic radius of Mg²⁺ allows water molecules to approach closer to the ion, further strengthening the ion-dipole interactions. Thus, the enthalpy change of hydration is more exothermic for Mg²⁺ due to its higher charge density and stronger interactions with water molecules compared to Na⁺.

