In biotechnology and bioengineering, enzymes are pivotal. They catalyse reactions in both biological systems and industrial processes. This section delves into the distinctions and applications of immobilised and free enzymes.
Introduction to Immobilised and Free Enzymes
Enzymes, as biocatalysts, facilitate and accelerate biochemical reactions. Immobilised enzymes are attached to a solid support or matrix, allowing them to be contained within a defined area while retaining their activity. They can be used repeatedly, a stark contrast to free enzymes, which are soluble and free to diffuse in the reaction medium.
Advantages of Immobilised Enzymes
Enhanced Stability
- Thermal Stability: Immobilised enzymes exhibit greater resistance to thermal denaturation. This means they can maintain their activity at higher temperatures, extending their use in processes where temperature fluctuations are common.
- pH Stability: They demonstrate a wider range of pH tolerance. This versatility is crucial in industrial applications where pH can vary.
Reusability
- Cost-Effectiveness: The reusability of immobilised enzymes translates to lower costs over time, as fewer enzymes need to be added to successive reactions.
- Operational Stability: They offer consistent activity over multiple uses, essential for continuous industrial processes.
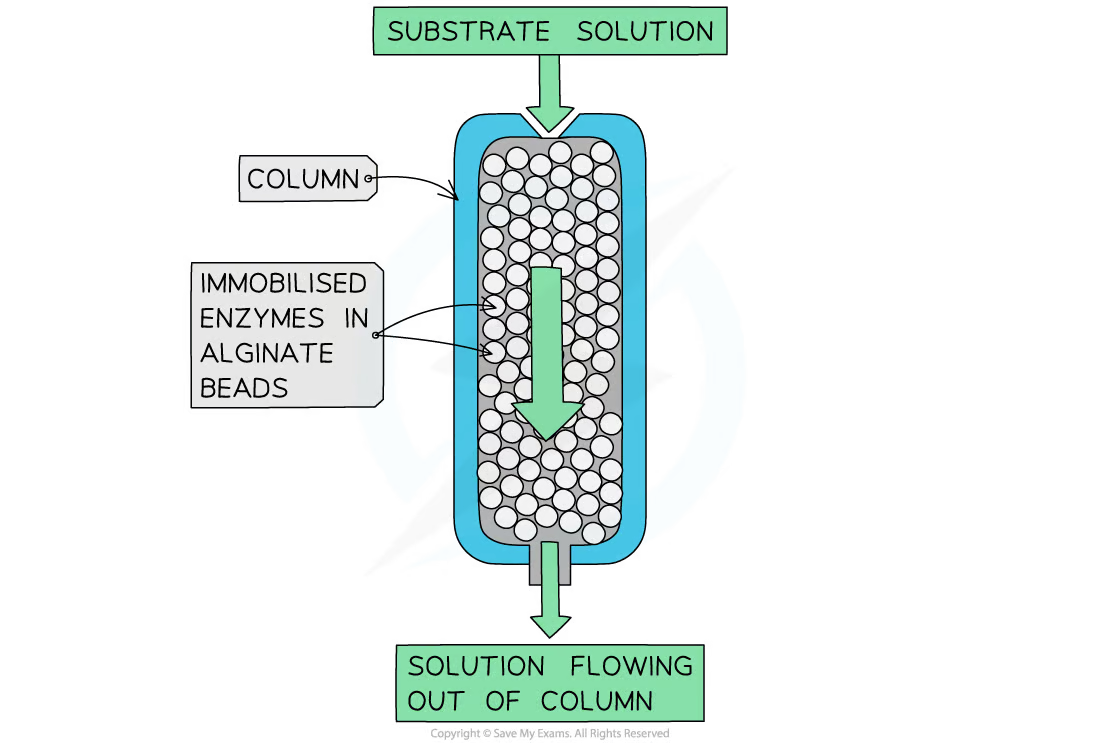
Image courtesy of Save My Exams
Controlled Reaction Conditions
- Easy Separation: Post-reaction, immobilised enzymes can be easily separated from the product, streamlining the purification process.
- Reduced Reaction Time: Due to higher local enzyme concentrations, reactions often proceed more rapidly.
Methods of Enzyme Immobilisation
Adsorption
- Principle: This method involves passive adsorption of enzymes onto surfaces like silica gel, activated carbon, or alumina.
- Advantages: It's a simple, cost-effective process requiring minimal enzyme modification.
- Limitations: Potential for enzyme leakage due to weak binding forces.
Covalent Bonding
- Principle: Covalently attaching enzymes to supports such as cellulose or glass beads ensures a strong, stable linkage.
- Advantages: Reduced enzyme leaching due to strong bonds.
- Challenges: The process can be complex and may affect the active site of the enzyme.
Entrapment
- Principle: Enzymes are entrapped within a polymer matrix or gel, like calcium alginate beads.
- Advantages: Provides a protective environment for enzymes, shielding them from harsh external conditions.
- Drawbacks: Potential diffusional limitations for substrates and products.
Cross-Linking
- Principle: Involves creating cross-links between enzyme molecules using agents like glutaraldehyde.
- Advantages: Enhances mechanical stability and can lead to high enzyme loading.
- Considerations: Can potentially alter the enzyme's active site.
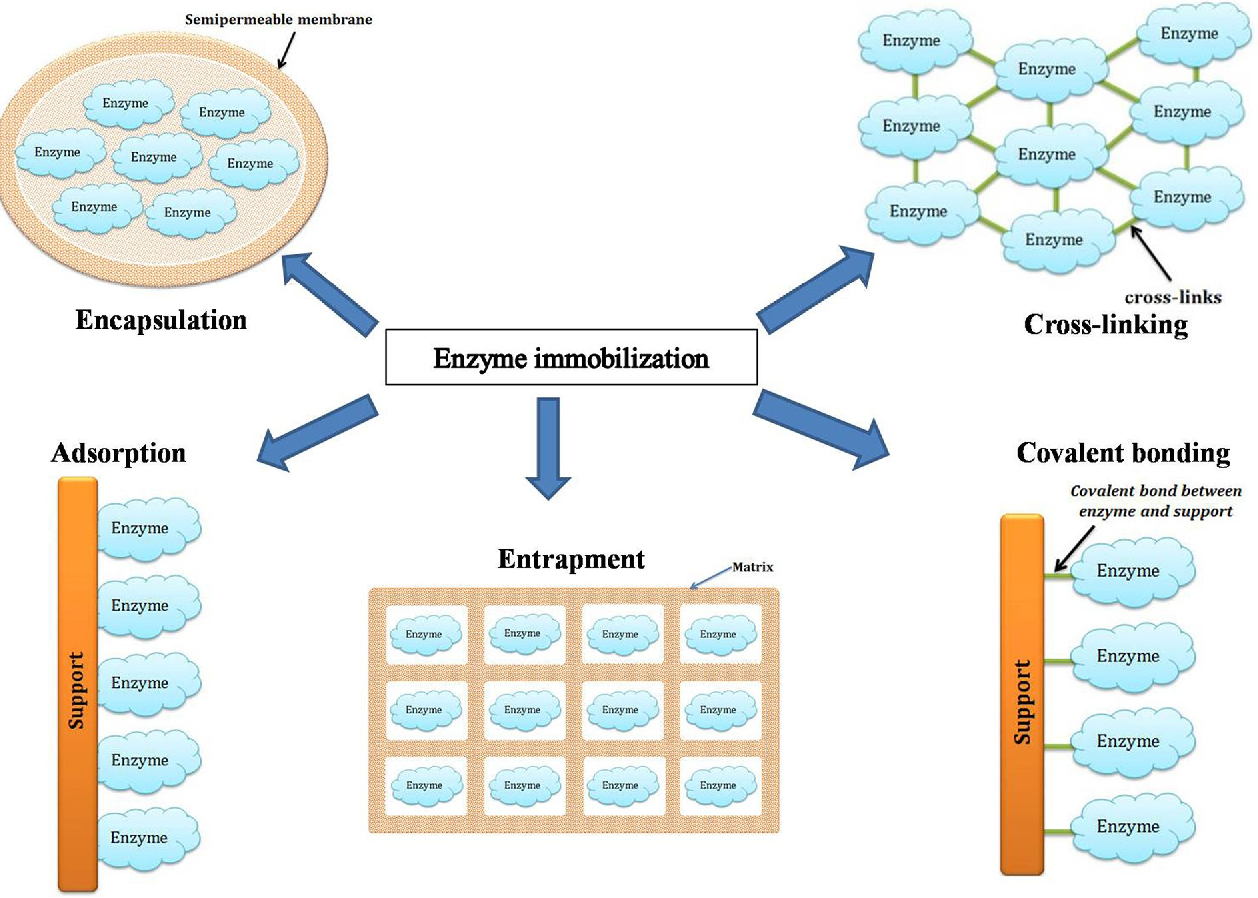
Image courtesy of Semantic Scholar
Practical Applications of Immobilised Enzymes
Bioreactors
- Waste Treatment: Employed in the treatment of wastewater, where they help in breaking down organic pollutants.
- Biofuel Production: Catalysts in the conversion process of biomass to bioethanol and biodiesel.
Medical Applications
- Diagnostics: Integral in biosensors for the detection of various biomolecules, crucial in medical diagnostics.
- Therapeutics: Used in controlled drug delivery systems for targeted treatment, enhancing efficacy and reducing side effects.
Industrial Processes
- Food Industry: Utilised in refining sugar, clarifying fruit juices, and synthesising flavour enhancers.
- Pharmaceuticals: Critical in producing semi-synthetic antibiotics, cholesterol-lowering drugs, and various pharmaceutical intermediates.
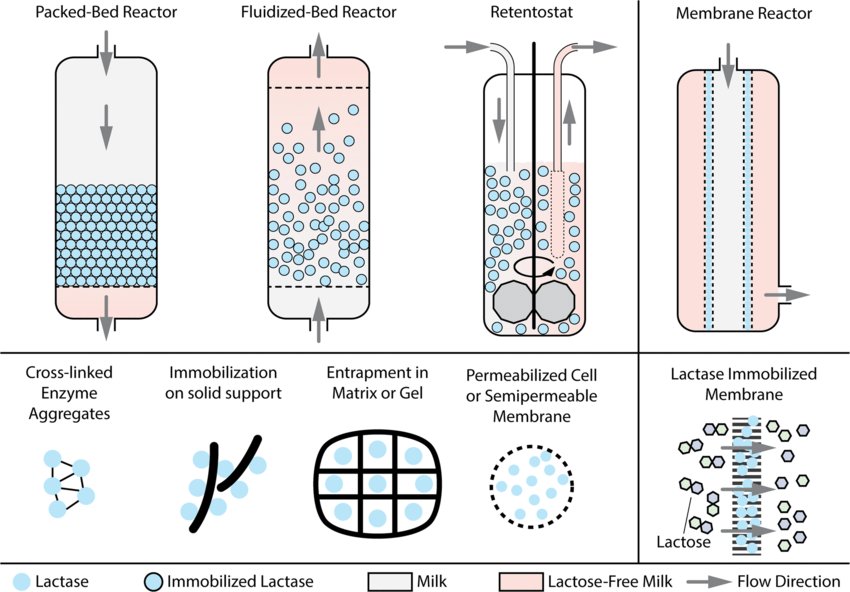
Image courtesy of ResearchGate
Comparison with Free Enzymes
Activity and Specificity
- Substrate Specificity: While immobilised enzymes may show different substrate specificity due to alterations in their three-dimensional structure, they generally retain the ability to catalyse the same reactions as their free counterparts.
- Activity Levels: Some immobilised enzymes experience reduced activity due to limited substrate access or conformational changes. However, this is often offset by their enhanced stability and reusability.
Application Suitability
- Free Enzymes: Preferred in small-scale, precision laboratory experiments. Ideal for processes where enzyme recovery is not necessary or where absolute enzyme activity is paramount.
- Immobilised Enzymes: Best suited for large-scale, continuous industrial processes where enzyme recovery and reuse are beneficial. They're also ideal in applications requiring controlled reaction conditions.
Economic Considerations
- Cost-Effectiveness: Initial costs for immobilisation can be high, but long-term savings are significant due to reduced enzyme consumption and simpler product purification processes.
- Process Efficiency: Immobilised enzymes can lead to more efficient processes by reducing reaction times and simplifying separation and purification steps.
Industrial and Environmental Implications
Sustainability
- Resource Efficiency: Using immobilised enzymes contributes to more sustainable processes by reducing waste and energy consumption.
- Environmental Impact: These enzymes reduce the environmental footprint of industrial processes by lowering the need for harsh chemicals and conditions.
Technological Innovations
- Emerging Techniques: Advances in nanotechnology and materials science are leading to more efficient and effective methods of enzyme immobilisation.
- Customisation: Tailoring immobilisation techniques to specific enzymes and processes is an area of ongoing research, promising more targeted and efficient applications.
In conclusion, immobilised enzymes offer significant advantages over free enzymes, particularly in industrial and environmental applications. Their enhanced stability, reusability, and ease of separation make them ideal for large-scale and continuous processes. Understanding these distinctions is crucial for students in the field of biotechnology, equipping them with the knowledge needed for future innovations in enzyme technology.
FAQ
Yes, immobilised enzymes are used in medical diagnostics, particularly in biosensors. These biosensors utilise immobilised enzymes to detect specific substances in biological samples, such as blood or urine. For instance, glucose biosensors, which are widely used in managing diabetes, employ immobilised glucose oxidase to detect glucose levels. The enzyme reacts with glucose in the sample, producing a measurable signal proportional to the glucose concentration. The use of immobilised enzymes in these sensors enhances their stability and allows for repeated use, making them practical for regular monitoring of various biomarkers.
In environmental biotechnology, immobilised enzymes play a significant role in pollution control and waste management. They are used in bioreactors for wastewater treatment, where they help in degrading toxic substances and organic pollutants, converting them into less harmful compounds. This application is especially valuable in treating industrial effluents that contain complex and hazardous chemicals. The immobilisation of enzymes in these systems enhances their stability and allows for their reuse, making the process more efficient and cost-effective. Furthermore, the use of enzymes in this context is environmentally friendly, as it avoids the need for harsh chemicals and extreme conditions.
The main challenges in enzyme immobilisation include maintaining enzyme activity, preventing enzyme leakage, and ensuring effective substrate access to the immobilised enzyme. Maintaining enzyme activity can be challenging as the immobilisation process can alter the enzyme’s structure and functionality. This issue is addressed by carefully selecting immobilisation techniques and materials that minimise alterations to the enzyme's active site. Preventing enzyme leakage is crucial for the stability and efficiency of the process. This is often managed by using stronger binding methods like covalent bonding. Ensuring effective substrate access involves designing the immobilisation matrix to have an optimal pore size and surface area, allowing easy diffusion of substrates and products to and from the enzyme.
Common materials used for immobilising enzymes include natural polymers like alginate and chitosan, synthetic polymers like polyacrylamide, and inorganic materials like silica gel and glass beads. These materials are chosen based on their biocompatibility, chemical stability, and ability to interact with enzymes either physically or chemically. Natural polymers are often preferred for their biocompatibility and gentle interaction with enzymes, which helps to retain enzymatic activity. Synthetic polymers and inorganic materials offer robustness and can be engineered to provide specific surface properties for enzyme attachment. The choice of material depends on the desired application and the specific requirements of the enzyme and process.
The immobilisation of enzymes can alter their kinetic properties like Vmax (maximum velocity) and Km (Michaelis constant). Often, immobilised enzymes exhibit a lower Vmax compared to their free counterparts. This decrease in Vmax is usually due to diffusional limitations, as the substrate must diffuse through the immobilising matrix to reach the enzyme. Additionally, changes in enzyme conformation upon immobilisation can affect substrate binding, which might increase the Km value, indicating a lower affinity for the substrate. However, it's important to note that these changes are highly dependent on the method of immobilisation and the nature of the enzyme and substrate involved.
Practice Questions
Immobilised enzymes offer enhanced thermal and pH stability compared to free enzymes. This means they can function effectively under a wider range of temperatures and pH levels, which is advantageous in industrial processes where such conditions can vary. Additionally, immobilised enzymes can be reused multiple times, making them more cost-effective. Their reusability not only reduces the frequency of enzyme replacement but also simplifies the purification process of the end products, as the enzymes can be easily separated from the reaction mixture.
One method of enzyme immobilisation is adsorption, where enzymes are passively adsorbed onto surfaces like activated carbon or silica gel. This method is advantageous due to its simplicity and cost-effectiveness. It requires minimal modification of the enzyme, thereby preserving its activity. However, a limitation of adsorption is the potential for enzyme leakage, as the binding forces between the enzyme and the support are often weak. This can lead to a gradual loss of enzyme from the support material, reducing the efficiency of the immobilisation over time and possibly contaminating the product with enzymes.

