Measuring enzyme activity is integral to understanding the roles and functions of enzymes in biological systems. This comprehensive guide delves into the methodologies for quantifying enzyme activity, with a specific focus on the measurement of product formation using catalase and substrate disappearance using amylase. It also includes a detailed discussion on the selection of appropriate assays and their limitations, providing an essential resource for A-Level Biology students.
1. Introduction to Enzyme Activity Measurement
Enzymes, as biological catalysts, are fundamental in driving biochemical reactions. The measurement of enzyme activity is a key aspect in biochemistry, allowing us to understand and quantify the rate at which enzymes catalyse reactions. This involves determining the speed of substrate conversion to product or the rate of substrate consumption, crucial for enzyme kinetics studies and various applications in research and industry.
2. Methodologies for Quantifying Enzyme Activity
2.1 Product Formation Using Catalase
2.1.1 Understanding Catalase
Catalase is an enzyme that accelerates the decomposition of hydrogen peroxide into water and oxygen, a reaction vital in protecting cells from oxidative damage. Due to its rapid reaction rate and the simplicity of observing its products, catalase is a prime candidate for studying enzyme kinetics.
2.1.2 Techniques for Measuring Catalase Activity
- Direct Measurement of Oxygen Production: This method involves collecting the oxygen produced in a graduated cylinder or gas syringe, providing a direct measurement of the enzyme's activity.
- Spectrophotometric Analysis: This approach utilises the decrease in hydrogen peroxide's absorbance at a specific wavelength to quantify enzyme activity. It's a widely used method due to its precision and convenience.
- Manometric Techniques: These techniques measure the pressure change due to oxygen production, offering a sensitive way to detect changes in catalase activity.
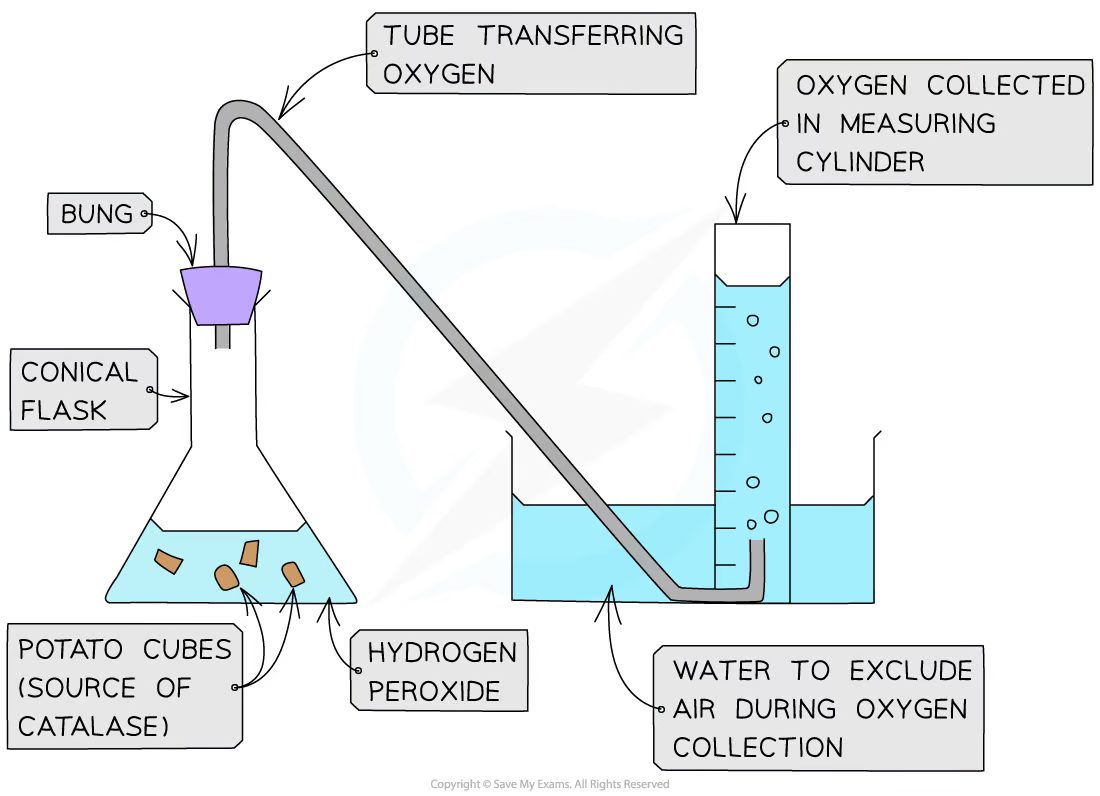
Image courtesy of Save My Exams
2.2 Substrate Disappearance Using Amylase
2.2.1 The Role of Amylase
Amylase plays a critical role in the digestive system by breaking down starch into simpler sugars. It's also significant in various industrial processes, making its study vital.
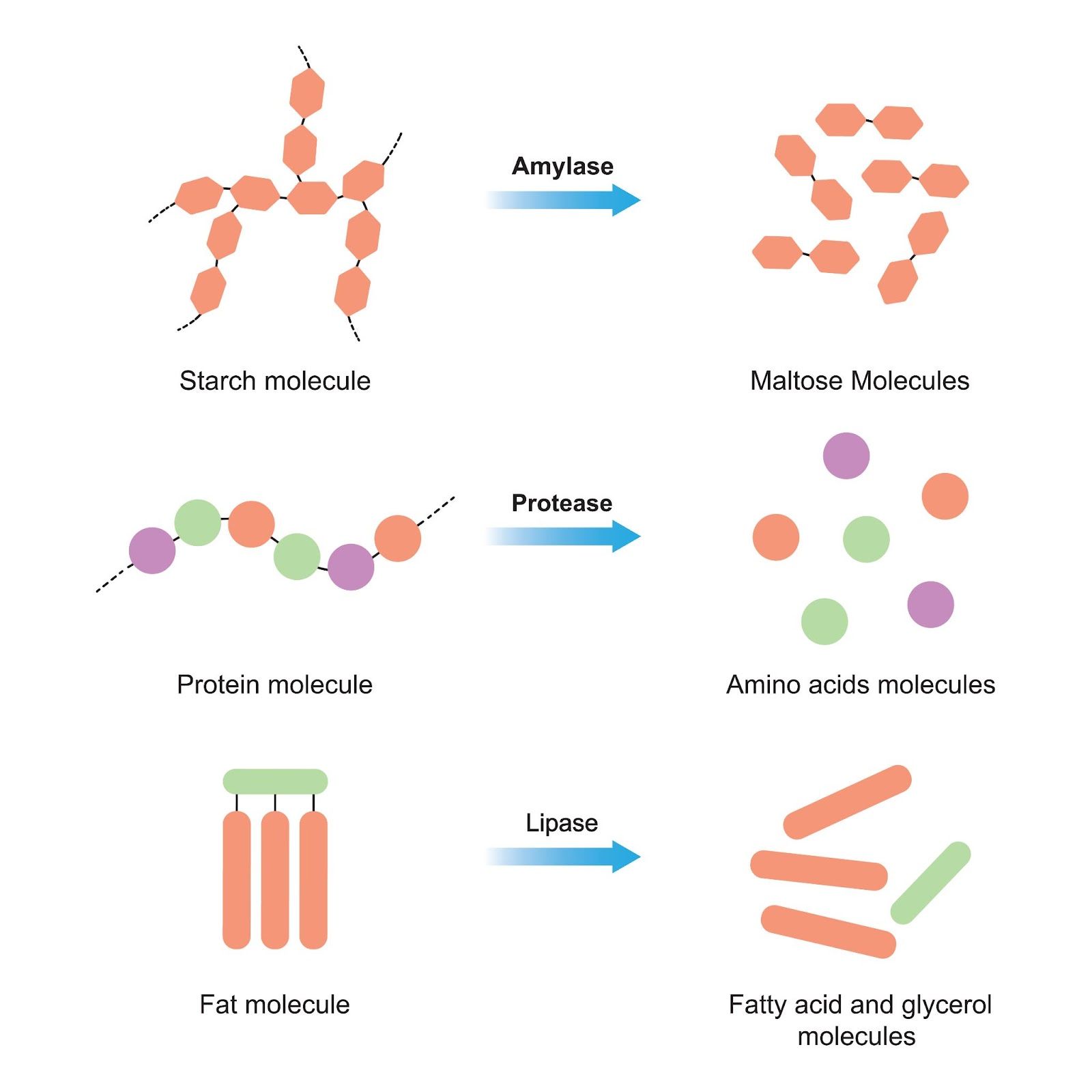
Image courtesy of Ali
2.2.2 Methods for Amylase Activity Measurement
- Iodine-Starch Complex Test: This visual assay involves adding iodine to the reaction mixture. The presence of starch creates a blue-black complex, which disappears as amylase breaks down the starch.
- Chromatographic Techniques: These allow for the separation and identification of sugars resulting from starch breakdown. It's a more sophisticated method, providing detailed insights into the enzymatic activity.
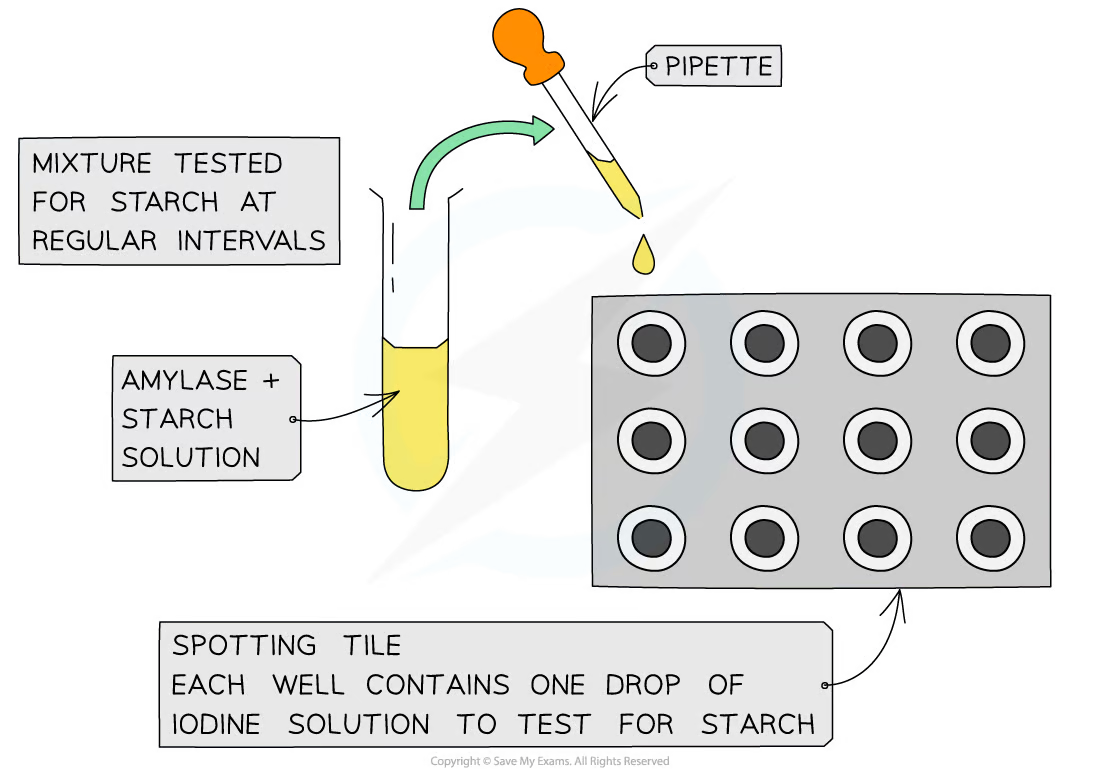
Image courtesy of Save My Exams
3. Selecting Appropriate Assays
3.1 Considerations in Assay Selection
- Specificity and Sensitivity: The chosen assay must specifically target the enzyme's activity and be sensitive enough to detect even small changes.
- Practicality and Accessibility: Factors like the availability of equipment, cost, and ease of execution play a significant role in assay selection.
- Reproducibility and Reliability: An ideal assay should produce consistent and reliable results under similar conditions.
3.2 Limitations of Current Assays
- Interference Issues: Other substances in the sample may interfere with the assay, leading to skewed results.
- Equipment Limitations: The accuracy of the results is partly dependent on the precision of the instruments used.
- Environmental Influences: External factors like temperature, pH, and ionic strength can affect the enzyme's activity, thus influencing the assay results.
4. Practical Aspects of Measuring Enzyme Activity
4.1 Sample Preparation
Proper sample preparation is vital. This involves adjusting environmental conditions to optimal levels for the specific enzyme being studied, ensuring that the enzyme retains its activity during the assay.
4.2 Calibration and Standardisation
For the reliability of results, calibration of equipment and standardisation of procedures are essential. This includes creating standard curves for comparison and ensuring the equipment, like spectrophotometers, is accurately calibrated.
4.3 Data Interpretation
Analysing the data from enzyme assays requires a thorough understanding of enzyme kinetics. Calculating reaction rates, comprehending enzyme behaviour, and interpreting results in biological contexts are integral to drawing accurate conclusions from the assays.
4.4 Ethical and Safety Considerations
When conducting enzyme assays, it's important to consider ethical implications, particularly if the enzymes or substrates are derived from living organisms. Additionally, laboratory safety, especially when handling potentially harmful substances like hydrogen peroxide, should always be a priority.
4.5 Technological Advancements
Recent advancements in technology have led to more sophisticated methods for measuring enzyme activity, including high-throughput screening and digital imaging techniques. These advancements offer greater accuracy and efficiency in enzyme activity measurement.
5. Challenges and Future Directions
5.1 Overcoming Limitations
Addressing the limitations of current assays, such as interference and sensitivity issues, is an ongoing challenge. Research is focused on developing more specific and sensitive assays.
5.2 Emerging Techniques
Emerging techniques, like molecular docking and computational modelling, are providing new ways to study enzyme activity. These methods offer insights into enzyme-substrate interactions at a molecular level.
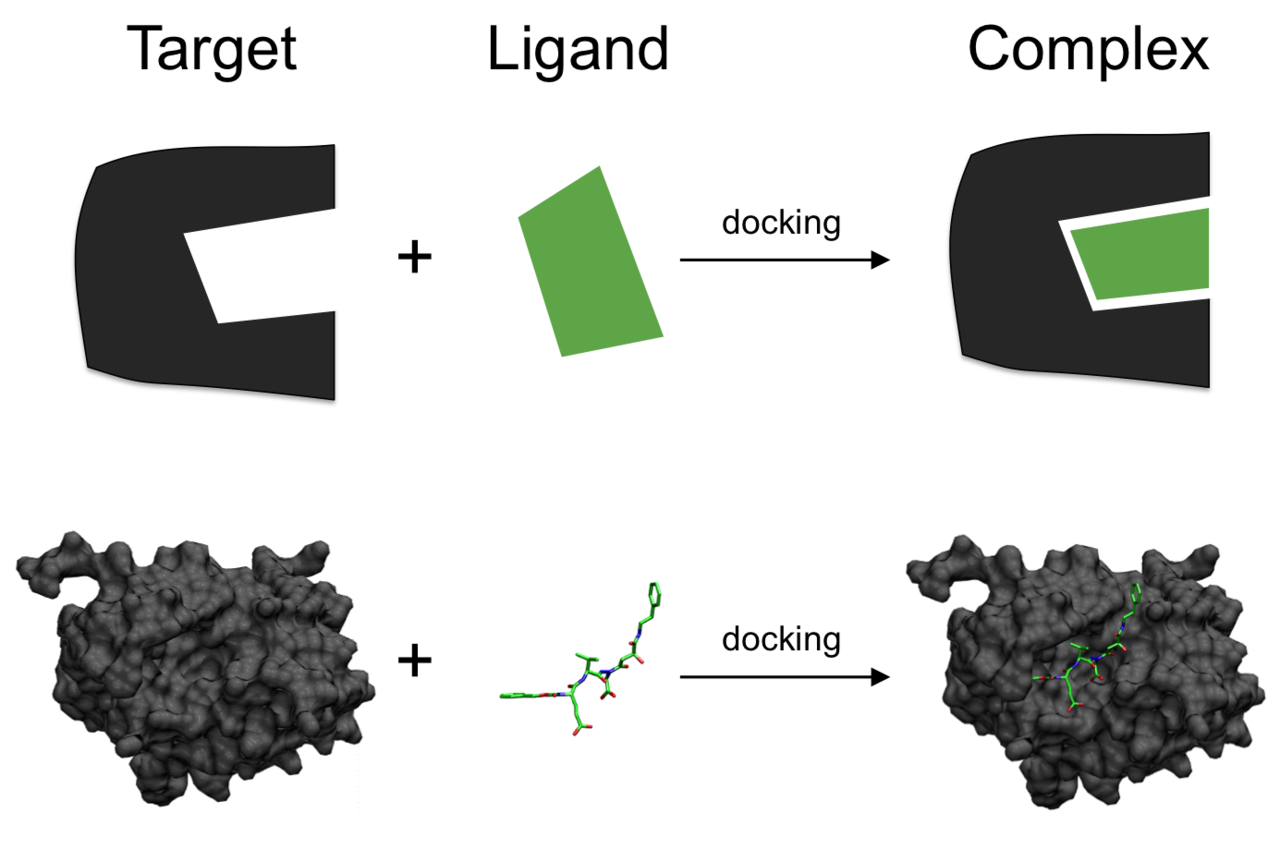
Schematic illustration of molecular docking
Image courtesy of Scigenis
5.3 Interdisciplinary Approaches
Combining knowledge from different fields, such as bioinformatics and nanotechnology, is opening up new avenues for measuring and manipulating enzyme activity. This interdisciplinary approach is crucial for the future of enzyme research.
In summary, measuring enzyme activity is a complex but essential aspect of studying biological systems. The methodologies, considerations, and advancements discussed here provide a thorough understanding of how enzyme activity is measured and the factors influencing these measurements. For students of A-Level Biology, grasping these concepts is fundamental to understanding the broader implications of enzymes in both biological and industrial contexts.
FAQ
Using a control in enzyme activity assays is crucial for validating the results. A control, typically a setup where the enzyme is absent or inactive, provides a baseline against which the changes observed in the experimental setup can be compared. This comparison helps in determining whether the observed effects are due to the enzyme's activity or other external factors. For instance, in a catalase activity assay, a control without catalase would indicate any changes in hydrogen peroxide concentration not caused by the enzyme. Controls are essential for scientific accuracy, ensuring that the results are due to the variable being tested and not due to other uncontrolled factors.
Yes, enzyme activity can be inhibited, which would significantly affect assay results. Inhibitors are substances that decrease the rate of enzyme-catalysed reactions. There are different types of inhibitors, such as competitive, non-competitive, and uncompetitive inhibitors. Competitive inhibitors compete with the substrate for the active site, while non-competitive inhibitors bind to another part of the enzyme, changing its shape and affecting its function. Uncompetitive inhibitors bind only to the enzyme-substrate complex. The presence of inhibitors in an enzyme assay can lead to a decreased reaction rate, which can be mistaken for a decrease in enzyme activity. Therefore, it's important to consider the presence of inhibitors when interpreting assay results.
Cofactors and coenzymes are non-protein molecules that assist enzymes in catalysing reactions. Cofactors, which can be inorganic (like metal ions) or organic molecules, often act as catalysts themselves or stabilise the enzyme structure. Coenzymes, a subset of cofactors, are organic molecules that transfer chemical groups from one molecule to another in a reaction. Their presence is essential for the proper functioning of many enzymes. In enzyme activity assays, the absence of necessary cofactors or coenzymes can result in decreased or absent enzyme activity. Therefore, ensuring the availability of these molecules in assays is crucial for obtaining accurate measurements of enzyme activity.
Temperature significantly impacts enzyme activity, thus affecting measurements in enzyme assays. Enzymes, being proteins, have an optimal temperature range where they function most efficiently. Above or below this range, the enzyme's structure can be altered, affecting its ability to catalyse reactions. In the case of catalase and amylase, a deviation from their optimal temperature can lead to a decreased reaction rate. High temperatures can cause denaturation, where the enzyme loses its structure and functionality, while low temperatures can slow down the reaction rate. Therefore, maintaining a consistent and optimal temperature is crucial in enzyme assays to ensure accurate measurements of enzyme activity.
Enzyme kinetics is pivotal in comprehending enzyme activity as it provides insights into the rate at which reactions occur and how they are affected by various factors. Understanding enzyme kinetics involves studying how changes in conditions like substrate concentration, pH, and temperature influence the rate of the enzymatic reactions. For instance, the Michaelis-Menten equation, a cornerstone of enzyme kinetics, describes the relationship between substrate concentration and reaction rate, helping to determine the maximum rate of the reaction (Vmax) and the substrate concentration at which the reaction rate is half of Vmax (Km). This information is crucial in understanding how enzymes function in different physiological environments and can aid in the design of drugs and in the industrial application of enzymes.
Practice Questions
The spectrophotometric method for measuring catalase activity is based on monitoring the decrease in absorbance of hydrogen peroxide at a specific wavelength. Catalase catalyses the breakdown of hydrogen peroxide into water and oxygen, reducing its concentration. A spectrophotometer measures the absorbance, which is inversely proportional to the concentration of hydrogen peroxide. Therefore, as catalase activity increases, the absorbance decreases. One limitation of this method is that it can be influenced by other light-absorbing substances in the sample, which can lead to inaccuracies in the results.
The iodine-starch test is used to measure amylase activity by adding iodine to a starch solution that is being acted upon by amylase. Amylase breaks down starch into simpler sugars, reducing the starch concentration in the solution. When iodine is added to starch, it forms a blue-black complex; as the starch concentration decreases due to amylase activity, the colour changes from blue-black to lighter or colourless. One factor affecting the accuracy of this test is the timing of iodine addition. If iodine is added too early or too late, it can lead to a misrepresentation of the actual amylase activity.

